Twisting the Knife: The Open Case of Menstruation, the Brain, and the Missed Disease of Endometriosis
Zoe Shelley
Illustrations by Abigail Schoenecker
Twisting the Knife: The Open Case of Menstruation, the Brain, and the Missed Disease of Endometriosis
Last night, you went to bed on time. But when you wake up today, you feel exhausted. Everywhere. Normally, you spring out of bed, throw on your favorite workout clothes, and are out and back from a run in no time. Instead, your muscles ache incessantly as you press yourself out of bed; every movement is in slow motion. And although your body feels stalled, your thoughts are speeding through your mind. An anxiety you rarely experience is now wreaking havoc, and a tightness forms in your shoulders and neck. Both are signs that an insistent buzz of stress has begun to settle in. On top of the hum of your racing thoughts, there is a relentless ache in your stomach. Even as you stand up and start to get ready for the day, the pain persists, cutting into every moment of your morning. The discomfort keeps you in bed for most of the day, a heating pad pressed onto your stomach. As you fall asleep that night, you think that maybe you are being hit with the worst case of the flu you have ever had. Awoken at 3:00 a.m. by the pang of your bladder calling you to the bathroom, you sit down and find your pajama pants have been stained with bright red blood. How could you have forgotten? Your period was due. This vicious cycle of events is a common occurrence for women everywhere. But now imagine that exhaustion, that irritability, that incessant, pulsing, never-ending pain, as not just a monthly factor of your life, but a near constant presence marking every day. This ceaseless pain is a reality for women living with endometriosis.
The Scene of the Crime: The Menstrual Cycle
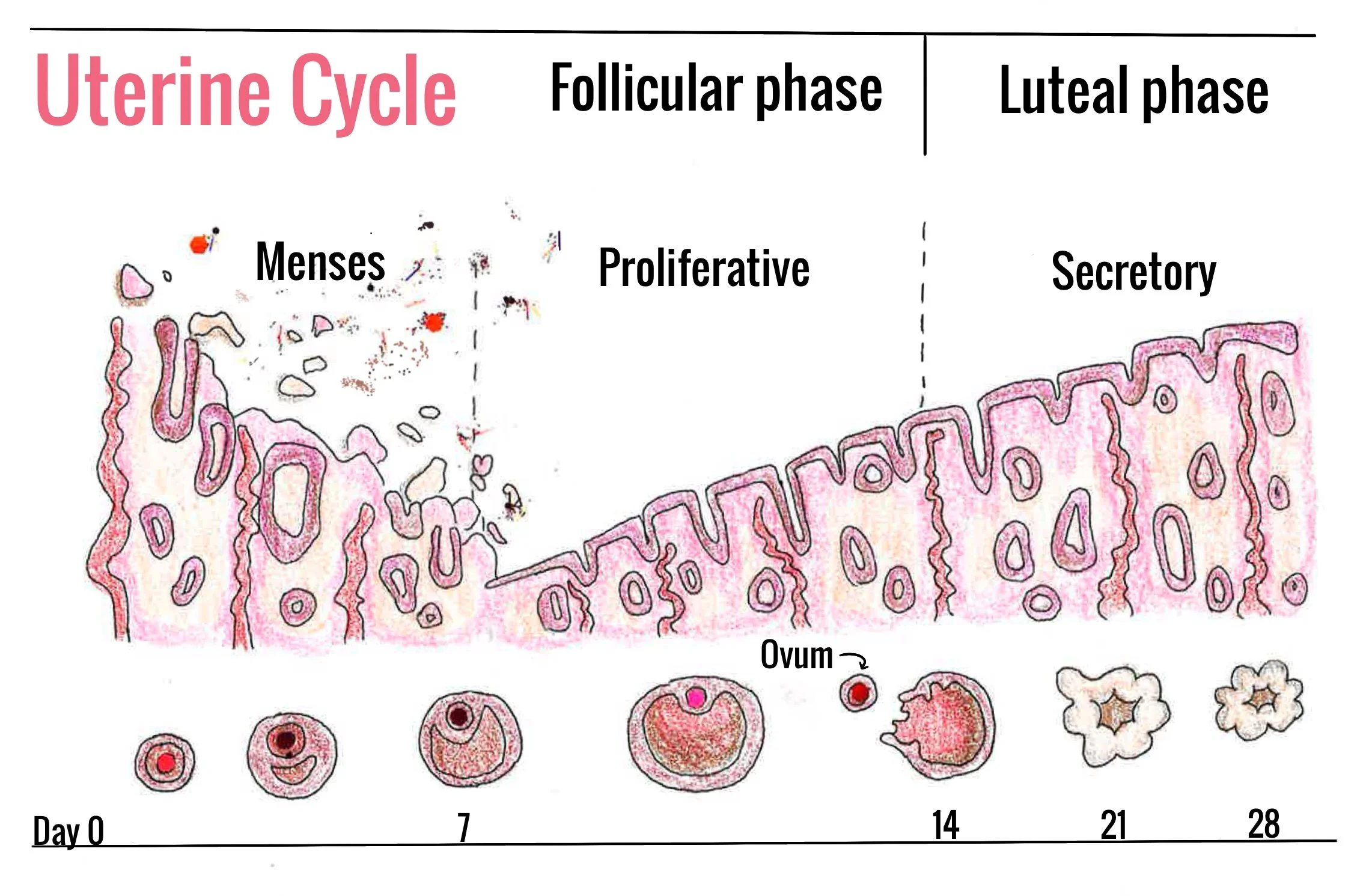
There are 1.8 billion people worldwide who menstruate every month [1]. In essence, menstruation is the shedding of the endometrium — the inner lining of the uterus, which is the central organ of the female reproductive system [2, 3]. During the menstrual cycle, the endometrium prepares for possible egg implantation, supports the uterus for pregnancy, and sheds in the absence of implantation [3]. All of these functions are regulated by chemical messengers that travel through the bloodstream called hormones [3]. A central hormone-regulating pathway is the hypothalamic-pituitary-ovarian (HPO) axis, which facilitates communication between the brain and the ovaries to orchestrate the menstrual cycle [4, 5]. The HPO axis begins with the hypothalamus, a brain region that controls a plethora of functions such as sleep, body temperature, and hunger — all of which keep our bodies in a balanced state [6]. The hypothalamus kicks off these processes by producing gonadotropin-releasing hormone (GnRH), which signals the pituitary gland — a major regulator of other hormone-secreting tissues — to secrete its own regulatory hormones called gonadotropins into the bloodstream [5]. These pituitary regulatory hormones are what truly drive the reproductive cycle [7]. The two gonadotropins secreted are luteinizing hormone (LH) and follicle-stimulating hormone (FSH) [7, 8]. A high enough concentration of LH and FSH cues the ovaries to secrete the hormones estrogen and progesterone [9, 10]. Estrogen and progesterone facilitate the growth of the endometrium and biological processes such as fertility and pregnancy [11]. These hormones signal back to the hypothalamus and the pituitary gland as needed, completing the cycle by releasing more GnRH, LH, or FSH [12]. Throughout the menstrual cycle, LH and FSH are released by the pituitary gland in an alternating pattern, dictating the phase of the cycle [7].
The menstrual cycle involves changes to the endometrium regulated by hormone fluctuations, and it is characterized by four phases: the follicular, ovulatory, luteal, and menstrual phases [13]. During the follicular phase, low levels of estrogen and progesterone prompt the pituitary gland to increase FSH secretion [14, 15]. As the pituitary gland releases more FSH, an immature egg starts to grow in the ovaries and releases estrogen, which stimulates the endometrium to thicken [14, 15 16]. In the ovulatory phase, estrogen and LH increase rapidly until they reach their peak [14]. Due to the surge in LH levels, the ovaries release the now mature egg into the fallopian tubes, part of the female reproductive system that connects the ovaries to the uterus [14, 17]. Progesterone levels rise in the middle of ovulation as LH surges [14, 18]. During the luteal phase, progesterone reaches its peak concentration before decreasing along with estrogen, and LH and FSH levels return to baseline. These hormonal changes trigger the menstrual phase, during which the thickened endometrium sheds and produces the bleeding we know as a period [14, 18]. By shedding resource-hungry tissue rather than maintaining it, the uterus can preserve a healthy environment, save energy, and rebuild when a new fertilized egg is present [19]. Simultaneously, the oscillating hormones throughout the menstrual cycle also create a myriad of physical symptoms.
The Prime Suspect: Inflammation
Throbbing pain in the lower abdomen, also known as cramps, is the most prevalent symptom of the menstrual cycle, experienced by most people who menstruate [20]. Cramps are the result of the uterus contracting to shed the endometrium and begin with immune cells recognizing the damaged tissue of the endometrium, prompting an inflammatory response [21]. During inflammation, immune cells remove damaged cells and initiate the process of repairing tissue [22, 23]. Immune cells control inflammation by releasing small proteins called cytokines [24]. Cytokines can be either pro-inflammatory, meaning they increase inflammation, or anti-inflammatory, meaning they decrease inflammation [25]. Pro-inflammatory cytokines trigger the release of prostaglandins, which in turn amplify the effects of the cytokines [26]. Prostaglandins are hormone-like substances produced at sites of tissue damage, and ultimately play a key role in the commonly resulting pain and inflammation [27, 28]. Just before the luteal phase ends, progesterone rapidly decreases, which triggers the production of prostaglandins from the endometrium to facilitate its shedding [29]. Concurrently, prostaglandins prompt uterine contractions and significantly increase the sensitivity of nerve endings, leading to the pain associated with menstrual cramps [30]. The higher the amount of prostaglandins produced, the more severe the menstrual cramps [29]. However, cramps are not the only physical symptoms that occur during the menstrual cycle — significant changes are also happening in the brain as a result of hormonal activity.
The Murder Weapons: Estrogen and Progesterone
As previously mentioned, hormones play a critical role in dictating the menstrual cycle. Estrogen and progesterone can enter the brain and bind to their respective hormone receptor proteins to cause a cellular response [31, 32]. When estrogen interacts with its receptor on neurons — a type of brain cell — the hormone alters the cells’ critical method of communication: neurotransmission [33]. Neurons release small molecules called neurotransmitters that act on other neurons and allow for communication within the brain [34, 35]. The feelings of anxiety, exhaustion, and lack of motivation in the days leading up to a period may be symptoms resulting from fluctuating estrogen and progesterone levels as hormones modify concentrations of certain neurotransmitters [14]. Dopamine, serotonin, glutamate, and GABA are key neurotransmitters that bring about changes associated with respective phases of the menstrual cycle [36]. Dopamine modulates reinforcement and assists in the regulation of mood [37, 38]. When estradiol increases, it leads to higher dopamine concentrations, resulting in enhanced motivation, boosted mood, and increased reward perception [39]. Increased levels of estradiol also stimulate an increase in serotonin levels, which are associated with reduced anxiety, improved mood, and enhanced cognitive functioning [40, 41]. Estradiol also inhibits the protein that breaks down serotonin and increases the sensitivity of serotonin receptors [42]. When estradiol rises in the follicular and ovulatory phases, dopamine and serotonin levels simultaneously increase [36, 37, 43]. Following this initial rise in dopamine and serotonin, estradiol decreases and progesterone increases during the luteal and menstrual phases, leading to reduced dopamine and serotonin synthesis and subsequent negative feelings [43]. Estradiol also directly modulates glutamate, the primary excitatory neurotransmitter which makes a neuron more likely to release neurotransmitters [44, 45, 46]. Increased progesterone, however, is associated with decreased glutamate signaling and lower neuronal excitability and thus lower chances for a neuron to release neurotransmitters [47]. As such, progesterone increases concentrations of the primary inhibitoryneurotransmitter, GABA, which works to balance neurotransmission in the brain by reducing neuronal activity [47, 48].
The Bloody Truth: Endometriosis
In individuals with endometriosis, the mechanisms of a typical menstrual cycle are dysregulated. Endometriosis is a chronic inflammatory condition characterized by the growth of endometrial-like tissue outside the uterus, typically in the lower abdomen [37, 49]. To have a better sense of the disruption seen in endometriosis, imagine how painful it would be if a segment of your stomach tissue with the role of performing stomach-specific tasks began to grow in your eye. In its diagnosis, endometriosis is categorized by location and severity: superficial endometriosis, deep infiltrating endometriosis, and ovarian endometriotic cysts [37]. Superficial endometriosis appears on the surface of the tissue in the lower abdominal regions; deep infiltrating endometriosis involves scar tissue that extends into the muscle layer of the intestines, bladder, or other critical organs; and in ovarian endometrial cysts, sac-like structures filled with fluid or masses of skin form inside the ovaries [37, 50].
Endometrial-like tissue developing in areas external to the uterus functions as it would inside the uterus: growing, thickening, and shedding each month [51]. Therefore, individuals with endometriosis still experience the monthly renovation of the uterine lining, but this drastic transformation of endometrial-like tissue also occurs in various other regions of their body simultaneously [52]. Outside the uterus, broken-down tissue has no way to exit the body, often causing immense or even paralyzing pain, as well as inflammation and adhesions [53]. The proliferation of endometrial-like tissue in regions where it should not be found also creates heavy menstrual bleeding, painful sex, and, in some cases, infertility [54]. There is no clear explanation for the exact cause of this dysfunction in tissue growth [55]. A leading theory is retrograde menstruation — when menstrual blood containing endometrial cells flows backwards through the fallopian tubes into the peritoneal cavity, fluid-filled areas between abdominal organs [56, 57]. However, retrograde menstruation occurs in most people who menstruate, so it cannot be the only cause of endometriosis [58]. Factors such as immune system dysfunction, endometrial cells spreading via the bloodstream, or tissues transforming themselves into endometrial tissue are some other theories regarding the causes of endometriosis [57]. The endometrial-like tissue that migrates to various regions of the body contains estrogen receptors, as well as the ability to produce its own estrogen [59]. This means that the survival and growth of endometrial-like tissues, just like the endometrium, the brain, and other tissues, are heavily influenced by estrogen produced during the menstrual cycle [60]. In a person who menstruates without endometriosis, estrogen will rise during the follicular phase and peak during ovulation, eventually decreasing when progesterone is produced in the ovaries [7]. However, individuals with endometriosis experience progesterone resistance, meaning progesterone cannot play its regulatory role [61]. As a result, estrogen will overpower all other hormones and cause inflammation [61, 62].
The Suspect Strikes Again: Inflammation and Gray Matter in Endometriosis
Women with endometriosis also have higher levels of prostaglandins and cytokines as a result of this estrogen-caused inflammation, which are known to contribute to the survival and growth of endometrial-like scar tissue [63, 64]. Prostaglandins also promote the expression of proteins that synthesize estrogen around the tissue, which further stimulates its growth and the progression of the condition [65]. Endometrial-like scar tissue itself has a high concentration of inflammatory cells and cytokines, creating an inherently inflammatory environment [66]. This high concentration is due to the constant production of pro-inflammatory cytokines, which are a hallmark of the scar tissue present in endometriosis. Due to the cycle of pro-inflammatory cytokines releasing prostaglandins and prostaglandins enhancing cytokines, the tissue is chronically self-inflamed. This chronic self-inflammation further contributes to the growth and sustainment of the endometrial-like scar tissue in the body. Endometriosis is effectively a self-growing, self-sustaining, and self-progressive disorder [66]. Additionally, prostaglandins affect sensory neurons, increasing their sensitivity and contributing to chronic inflammatory pain symptoms [63]. This increase in inflammation and neuronal sensitivity leads to non-painful stimuli becoming painful [67]. Something as simple as a papercut can be excruciating when there is enough external inflammation and sensitivity. Thus, when the inflammation and sensitivity turn internal, any simple biological process — all of which involve internal stimuli — turns all the more excruciating [67].
Typically, inflammation is associated with reduced gray matter; however, this is not seen in endometriosis [68]. Gray matter is brain tissue made up of cell bodies, the ‘processing’ segment of the neuron [69]. Throughout a typical menstrual cycle, gray matter thickness fluctuates in response to estradiol and progesterone, although there is no conclusive research on the specific changes associated with each phase [70]. Ovarian hormones have been shown to cause gray matter increase or decrease depending on the region of the brain [70]. These standard gray matter volume changes that occur in all people who menstruate are also seen in individuals with endometriosis [70, 71]. But in comparison to people who menstruate without endometriosis, women who suffer from endometriosis may experience increased gray matter volume specifically in the cerebellum, a brain region critical to processing and anticipation of pain as well as pain-related emotional responses, which suggests that their pain processing is amplified [71, 72]. For people with endometriosis, the combined effects of changes to function and structure in the brain give rise to pain levels higher than the norm in the menstrual cycle.
Case Not Closed: Lack of Research Funding Caught Red Handed
Endometriosis was first identified as a disease more than a hundred years ago, but progress on finding treatments or even accessible diagnoses has since been tediously slow [73]. It was only in 1993 that the National Institute of Health passed a law allowing women to be studied in clinical trials, and it was only a year later that the Food and Drug Administration established the Office of Women’s Health, dedicated to ensuring the inclusion of women in clinical trials and the advancement of women’s health care [74]. Knowing this, it doesn’t come as a surprise that women will spend an average 25% more years in poor health than men [74]. Today, 10% of women worldwide have been diagnosed with endometriosis [54, 75]. Despite this condition's prevalence, it can take women 7–10 years to get a diagnosis, with women of color experiencing even longer delays [75, 76]. Considering the array of painful symptoms associated with endometriosis, waiting for a diagnosis is extremely detrimental to quality of life [71]. Additionally, delays in diagnosis make it difficult for individuals to find effective treatments [77]. This situation is exacerbated by the fact that treatment options are already limited due to not yet truly understanding the precise cause of endometriosis, which can be attributed to the lack of funding for research. Subsequently, the absence of funding results in less incentive for researchers to conduct further studies to fill knowledge gaps about the condition, preventing further advancements in treatment and diagnosis methods. In combination, these factors create a cycle of ignorance that further prolongs and worsens an already painful process of seeking relief for those with the disease [77]. Doctors and researchers must continue delving into the fascinating role estrogen and progesterone play in catalyzing changes in neurotransmitter concentrations, gray matter volumes, and the structure of brain regions. An improved comprehension of these mechanisms, spurred by adequate research funding, could ensure that all people who menstruate — especially those with endometriosis — are one step closer to understanding the full extent to which their menstrual cycle affects their bodies.
References
Rohatgi, A., & Dash, S. (2023). Period poverty and mental health of menstruators during COVID-19 pandemic: Lessons and implications for the future. Frontiers in Global Women’s Health, 4. doi: 10.3389/fgwh.2023.1128169
Watters, M., Martínez-Aguilar, R., & Maybin, J. A. (2022). The menstrual endometrium: From physiology to future treatments. Frontiers in Reproductive Health, 3. doi: 10.3389/frph.2021.794352
Critchley, H. O., Maybin, J. A., Armstrong, G. M., & Williams, A. R. (2020). Physiology of the endometrium and regulation of menstruation. Physiological Reviews, 100(3), 1149–1179. doi: 10.1152/physrev.00031.2019
Janes, L. (2020). Hypothalamic-pituitary-gonadal (HPG) axis. Encyclopedia of Personality and Individual Differences, 2112–2114. doi: 10.1007/978-3-319-24612-3_766
Zheng, C.-Y., Yu, Y.-X., Cao, S.-Y., & Bai, X. (2024). Epigenetics of inflammation in hypothalamus pituitary gonadal and neuroendocrine disorders. Seminars in Cell & Developmental Biology, 154, 340–345. doi: 10.1016/j.semcdb.2023.04.001
Xie, Y., & Dorsky, R. I. (2017). Development of the hypothalamus: Conservation, modification and Innovation. Development, 144(9), 1588–1599. doi: 10.1242/dev.139055
Herbison, A. E. (2020). A simple model of estrous cycle negative and positive feedback regulation of GnRH secretion. Frontiers in Neuroendocrinology, 57, 100837. doi: 10.1016/j.yfrne.2020.100837
Lambalk, C. B. (2023). The enigma of the gonadotropin-releasing hormone pulse frequency governing individual secretion of luteinizing hormone and follicle-stimulating hormone. F&S Reports, 4(2), 27–32. doi: 10.1016/j.xfre.2023.02.010
Oktem, O., Akin, N., Bildik, G., Yakin, K., Alper, E., Balaban, B., & Urman, B. (2017). FSH stimulation promotes progesterone synthesis and output from human granulosa cells without luteinization. Human Reproduction, 32(3), 643–652. doi: 10.1093/humrep/dex010
Allen, J. J., Herrick, S. L., & Fortune, J. E. (2016). Regulation of steroidogenesis in fetal bovine ovaries: Differential effects of LH and FSH. Journal of Molecular Endocrinology, 57(4), 275–286. doi: 10.1530/jme-16-0152
Dias Da Silva, I., Wuidar, V., Zielonka, M., & Pequeux, C. (2024). Unraveling the dynamics of estrogen and progesterone signaling in the endometrium: An overview. Cells, 13(15), 1236. doi: 10.3390/cells13151236
Kauffman, A. S. (2022). Neuroendocrine mechanisms underlying estrogen positive feedback and the LH surge. Frontiers in Neuroscience, 16. doi: 10.3389/fnins.2022.953252
Schmalenberger, K. M., Tauseef, H. A., Barone, J. C., Owens, S. A., Lieberman, L., Jarczok, M. N., Girdler, S. S., Kiesner, J., Ditzen, B., & Eisenlohr-Moul, T. A. (2021). How to study the menstrual cycle: Practical tools and recommendations. Psychoneuroendocrinology, 123, 104895. doi: 10.1016/j.psyneuen.2020.104895
Draper, C. F., Duisters, K., Weger, B., Chakrabarti, A., Harms, A. C., Brennan, L., Hankemeier, T., Goulet, L., Konz, T., Martin, F. P., Moco, S., & van der Greef, J. (2018). Menstrual cycle rhythmicity: Metabolic Patterns in healthy women. Scientific Reports, 8(1). doi: 10.1038/s41598-018-32647-0
Avila-Varela, D. S., Hidalgo-Lopez, E., Dagnino, P. C., Acero-Pousa, I., del Agua, E., Deco, G., Pletzer, B., & Escrichs, A. (2024). Whole-brain dynamics across the menstrual cycle: The role of hormonal fluctuations and age in healthy women. Npj Women’s Health, 2(1). doi: 10.1038/s44294-024-00012-4
Zhang, W. Y., McCracken, M., Dominguez, L. V., Zhang, A., Johal, J., & Aghajanova, L. (2024). The impact of estradiol supplementation on endometrial thickness and intrauterine insemination outcomes. Reproductive Biology, 24(2), 100886. doi: 10.1016/j.repbio.2024.100886
Makhanova, A., Maner, J. K., Joslin, M. D. M., Phimmasene, K. E. N., Jasienska, G., & Marcinkowska, U. M. (2025). Women’s affiliation across the menstrual cycle: Cycle phase variation and associations with progesterone and estradiol. Hormones and Behavior, 170, 105709. doi: 10.1016/j.yhbeh.2025.105709
Practice Committees of the American Society for Reproductive Medicine and the Society for Reproductive Endocrinology and Infertility. (2021). Diagnosis and treatment of luteal phase deficiency: A committee opinion. Fertility and Sterility, 115(6), 1416–1423. doi: 10.1016/j.fertnstert.2021.02.010
Okada, H., Tsuzuki, T., & Murata, H. (2018). Decidualization of the human endometrium. Reproductive Medicine and Biology, 17(3), 220–227. doi: 10.1002/rmb2.12088
Serrahima, C., & Martínez, M. (2023). The experience of dysmenorrhea. Synthese, 201(5). doi: 10.1007/s11229-023-04148-9
Kyathanahalli, C. N., Tu, F. F., Ashenafi, G., Schroer, M. S., & Hellman, K. M. (2025). Seeking the root causes of menstrual pain: A systematic review of biomarkers in menstrual effluent. Molecular Pain, 21. doi: 10.1177/17448069251360092
Chavda, V. P., Feehan, J., & Apostolopoulos, V. (2024). Inflammation: The cause of all diseases. Cells, 13(22), 1906. doi: 10.3390/cells13221906
Fang, X.-X., Zhai, M.-N., Zhu, M., He, C., Wang, H., Wang, J., & Zhang, Z.-J. (2023). Inflammation in pathogenesis of chronic pain: Foe and friend. Molecular Pain, 19. doi: 10.1177/17448069231178176
Liu, C., Chu, D., Kalantar‐Zadeh, K., George, J., Young, H. A., & Liu, G. (2021). Cytokines: From clinical significance to quantification. Advanced Science, 8(15). doi: 10.1002/advs.202004433
Liu, M., Saredy, J., Zhang, R., Shao, Y., Sun, Y., Yang, W. Y., Wang, J., Liu, L., Drummer, C., Johnson, C., Saaoud, F., Lu, Y., Xu, K., Li, L., Wang, X., Jiang, X., Wang, H., & Yang, X. (2020). Approaching inflammation paradoxes—proinflammatory cytokine blockages induce inflammatory regulators. Frontiers in Immunology, 11. doi: 10.3389/fimmu.2020.554301
Yao, C., & Narumiya, S. (2018). Prostaglandin‐cytokine crosstalk in chronic inflammation. British Journal of Pharmacology, 176(3), 337–354. doi: 10.1111/bph.14530
Jin, D., & Zhong, T. P. (2021). Prostaglandin signaling in ciliogenesis and development. Journal of Cellular Physiology, 237(6), 2632–2643. doi: 10.1002/jcp.30659
Tallima, H., & El Ridi, R. (2018). Arachidonic acid: Physiological roles and potential health benefits – A Review. Journal of Advanced Research, 11, 33–41. doi: 10.1016/j.jare.2017.11.004
Fajrin, I., Alam, G., & Usman, A. N. (2020). Prostaglandin level of primary dysmenorrhea pain sufferers. Enfermería Clínica, 30, 5–9. doi: 10.1016/j.enfcli.2019.07.016
Barcikowska, Z., Rajkowska-Labon, E., Grzybowska, M. E., Hansdorfer-Korzon, R., & Zorena, K. (2020). Inflammatory markers in dysmenorrhea and therapeutic options. International Journal of Environmental Research and Public Health, 17(4), 1191. doi: 10.3390/ijerph17041191
Pillerová, M., Borbélyová, V., Hodosy, J., Riljak, V., Renczés, E., Frick, K. M., & Tóthová, Ľ. (2021). On the role of sex steroids in biological functions by classical and non-classical pathways. an update. Frontiers in Neuroendocrinology, 62, 100926. doi: 10.1016/j.yfrne.2021.100926
Prior, J. C. (2020). Women’s reproductive system as balanced estradiol and progesterone actions—a revolutionary, paradigm-shifting concept in women’s health. Drug Discovery Today: Disease Models, 32, 31–40. doi: 10.1016/j.ddmod.2020.11.005
Lewitus, V. J., & Blackwell, K. T. (2023). Estradiol receptors inhibit long-term potentiation in the dorsomedial striatum. Eneuro, 10(8). doi: 10.1523/eneuro.0071-23.2023
Teleanu, R. I., Niculescu, A.-G., Roza, E., Vladâcenco, O., Grumezescu, A. M., & Teleanu, D. M. (2022). Neurotransmitters—key factors in neurological and neurodegenerative disorders of the Central Nervous System. International Journal of Molecular Sciences, 23(11), 5954. doi: 10.3390/ijms23115954
Rizo, J. (2018). Mechanism of neurotransmitter release coming into focus. Protein Science, 27(8), 1364–1391. doi: 10.1002/pro.3445
Del Río, J. P., Alliende, M. I., Molina, N., Serrano, F. G., Molina, S., & Vigil, P. (2018). Steroid hormones and their action in women’s brains: The importance of hormonal balance. Frontiers in Public Health, 6. doi: 10.3389/fpubh.2018.00141
Wang, J.-X., Zhuang, J.-Y., Fu, L., Lei, Q., Fan, M., & Zhang, W. (2020). How ovarian hormones influence the behavioral activation and inhibition system through the dopamine pathway. PLOS ONE, 15(8). doi: 10.1371/journal.pone.0237032
Pourhamzeh, M., Moravej, F. G., Arabi, M., Shahriari, E., Mehrabi, S., Ward, R., Ahadi, R., & Joghataei, M. T. (2021). The roles of serotonin in neuropsychiatric disorders. Cellular and Molecular Neurobiology, 42(6), 1671–1692. doi: 10.1007/s10571-021-01064-9
Diekhof, E. K. (2018). Estradiol and the reward system in humans. Current Opinion in Behavioral Sciences, 23, 58–64. doi: 10.1016/j.cobeha.2018.03.010
Azargoonjahromi, A., & Alzheimer’s Disease Neuroimaging Initiative. (2024). Serotonin enhances neurogenesis biomarkers, hippocampal volumes, and cognitive functions in alzheimer’s disease. Molecular Brain, 17(1). doi: 10.1186/s13041-024-01169-4
Quah, S. K. L., McIver, L., Roberts, A. C., & Santangelo, A. M. (2020). Trait anxiety mediated by amygdala serotonin transporter in the common marmoset. The Journal of Neuroscience, 40(24), 4739–4749. doi: 10.1523/jneurosci.2930-19.2020
Hernández-Hernández, O. T., Martínez-Mota, L., Herrera-Pérez, J. J., & Jiménez-Rubio, G. (2019). Role of estradiol in the expression of genes involved in serotonin neurotransmission: Implications for female depression. Current Neuropharmacology, 17(5), 459–471. doi: 10.2174/1570159x16666180628165107
E. Yoest, K., A. Cummings, J., & B. Becker, J. (2015). Estradiol, dopamine and motivation. Central Nervous System Agents in Medicinal Chemistry, 14(2), 83–89. doi: 10.2174/1871524914666141226103135
Rodríguez-Campuzano, A. G., & Ortega, A. (2021). Glutamate transporters: Critical components of glutamatergic transmission. Neuropharmacology, 192, 108602. doi: 10.1016/j.neuropharm.2021.108602
Mutluay, S. U., & Karataş, H. (2022). A review of glutamate and its receptors: Their roles in Brain Physiology and pathology. Acta Medica, 53(2), 99–109. doi: 10.32552/2022.actamedica.650
Teleanu, R. I., Niculescu, A.-G., Roza, E., Vladâcenco, O., Grumezescu, A. M., & Teleanu, D. M. (2022a). Neurotransmitters—key factors in neurological and neurodegenerative disorders of the Central Nervous System. International Journal of Molecular Sciences, 23(11), 5954. doi: 10.3390/ijms23115954
Bencker, C., Gschwandtner, L., Nayman, S., Grikšienė, R., Nguyen, B., Nater, U. M., Guennoun, R., Sundström-Poromaa, I., Pletzer, B., Bixo, M., & Comasco, E. (2025). Progestagens and progesterone receptor modulation: Effects on the brain, mood, stress, and cognition in females. Frontiers in Neuroendocrinology, 76, 101160. doi: 10.1016/j.yfrne.2024.101160
Jung, J., Williams, S., & Ralph, M. A. (2025). The Role of GABA in Semantic Memory and Its Neuroplasticity. doi: 10.7554/elife.91771.3
Charatsi, D., Koukoura, O., Gkorezi Ntavela, I., Chintziou, F., Gkorila, G., Tsagkoulis, M., Mikos, T., Pistofidis, G., Hajiioannou, J., Daponte, A. (2018). Gastrointestinal and Urinary Tract Endometriosis: A review on the Commonest Locations of Extrapelvic Endometriosis. Advances in Medicine, 2018(1). doi: 10.1155/2018/3461209
Rocha, T.P., Andres, M.P., Carmona, F., Baracat E.C., Abrão M.S. (2022). Deep Endometriosis: the Involvement of Multiple Pelvic Compartments Is Associated with More Severe Pain Symptoms and Infertility. Reproductive Sciences, 30, 1668–1675. doi: 10.1007/s43032-022-01104-9
Allaire, C., Bedaiwy, M.A., Yong, P.J. (2023). Diagnosis and management of endometriosis. Canadian Medical Association Journal, 195(10). doi: 10.1503/cmaj.220637
Saunders, P.T.K., Horne, A.W. (2021). Endometriosis: Etiology, pathobiology, and therapeutic prospects. Cell, 184(11), 2807-2824. doi: 10.1016/j.cell.2021.04.041
Gruber, T. M., Mechsner, S. (2021). Pathogenesis of Endometriosis: The Origin of Pain and Subfertility. Cells, 10(6), 1381. doi: 10.3390/cells10061381’
Hudson, N. (2021). The missed disease? Endometriosis as an example of 'undone science'. Reproductive Biomedicine & Society Online, 14, 20-27. doi: 10.1016/j.rbms.2021.07.003.
Cousins, F.L., McKinnon, B.D., Mortlock, S., Fitzgerald, H.C., Zhang, C., Montgomery, G.W., Gargett, C.E. (2023). New concepts on the etiology of endometriosis. Journal of Obstetrics and Gynaecology Research, 49(4). doi: 10.1111/jog.15549
Bermo, M.S., Koppula, B., Kumar, M., Leblond, A., Matesan, M.C. (2020). The Peritoneum: What Nuclear Radiologists Need to Know. Seminars in Nuclear Medicine, 50(5), 405-418. doi: 10.1053/j.semnuclmed.2020.04.005
Hegazy, A.A. (2024). International Journal of Reproductive BioMedicine. doi: 10.18502/ijrm.v22i5.16433
Viganò, P., Caprara, F., Giola, F., Di Stefano, G., Somigliana, E., Vercellini, P. (2024). Is retrograde menstruation a universal, recurrent, physiological phenomenon? A systematic review of the evidence in humans and non-human primates. Human Reproduction Open, 2024(3). doi: 10.1093/hropen/hoae045
Nie, J., Yi, Y., Liu, X., Guo, S.W. (2025). Progressively diminished estrogen signaling concordant with increased fibrosis in ectopic endometrium. Human Reproduction Open, 2025(3). doi: 10.1093/hropen/hoaf028
Garvey, M. (2024). Endometriosis: Future Biological Perspectives for Diagnosis and Treatment. International Journal of Molecular Sciences, 25(22). doi: 10.3390/ijms252212242.
Wang, Y., Nicholes, K., Shih, L.M. (2020). The Origin and Pathogenesis of Endometriosis. Annual Review Pathology: Mechanisms Disease, 15, 71-95. doi: 10.1146/annurev-pathmechdis-012419-032654
Oală, I.E., Mitranovici, M.I., Chiorean, D.M., Irimia, T., Crișan, A.I., Melinte, I.M., Cotruș, T., Tudorache, V., Moraru, L., Moraru, R., Caravia, L., Morariu, M., Pușcașiu, L. (2024). Endometriosis and the Role of Pro-Inflammatory and Anti-Inflammatory Cytokines in Pathophysiology: A Narrative Review of the Literature. Diagnostics,14(3), 312. doi: 10.3390/diagnostics14030312
Machairiotis, N., Vasilakaki, S., Thomakos, N. (2021). Inflammatory Mediators and Pain in Endometriosis: A Systematic Review. Biomedicines, 9(1), 54. doi: 10.3390/biomedicines9010054
Itani, R., Soubra, L., Karout, S., Rahme, D., Karout, L., Khojah, H.M.J. (2022). Primary Dysmenorrhea: Pathophysiology, Diagnosis, and Treatment Updates. Korean Journal of Family Medicine, 43(2), 101-108. doi: 10.4082/kjfm.21.0103.
Tamura, K., Yoshie, M., Kusama, K., Tsuru, A. (2025). Mechanisms of Decidual Dysfunction and Infertility in Endometriosis: Roles of Prostaglandins and SASP. Reproductive Medicine and Biology, 24(1). doi: 10.1002/rmb2.12663
Wang, L., Xing, Y. (2026). Exploring the therapeutic potential of disulfiram in endometriosis: mechanisms targeting inflammation, oxidative stress, pyroptosis, and angiogenesis. Gynecology and Obstetrics, 313. doi: 10.1007/s00404-026-08355-w
Jang, Y., Kim, M. & Hwang, S.W. (2020). Molecular mechanisms underlying the actions of arachidonic acid-derived prostaglandins on peripheral nociception. Journal of Neuroinflammation, 17. doi: 10.1186/s12974-020-1703-1
Zhang, Y., Catts, V., Sheedy, D., McCrossin, T., Kril, J.J., Weickert, C.S. (2016). Cortical grey matter volume reduction in people with schizophrenia is associated with neuro-inflammation. Translational Psychiatry, 6. doi: 10.1038/tp.2016.238
Aird-Rossiter, C., Zhang. H., Alexander, D.C., Jones, D.K., Palombo, M. (2026). Decoding gray matter, large-scale analysis of brain cell morphometry to inform microstructural modeling of diffusion MR signals. Communications Biology, 9(1), 138. doi: 10.1038/s42003-025-09353-5.
Rizor, E.J., Babenko, V., Dundon, N.M., Beverly-Aylwin, R., Stump, A., Hayes, M., Herschenfeld-Catalan. L., Jacobs, E.G., Grafton, S.T. (2024). Menstrual cycle-driven hormone concentrations co-fluctuate with white and gray matter architecture changes across the whole brain. Human Brain Mapping, 45(11). doi: 10.1002/hbm.26785.
Maulitz, L., Nehls, S., Stickeler, E., Ignatov, A., Kupec, T., Henn, A.T., Chechko, N., Tchaikovski, S.N. (2024). Psychological characteristics and structural brain changes in women with endometriosis and endometriosis-independent chronic pelvic pain. Human Reproduction, 39(11), 2473-2484. doi: 10.1093/humrep/deae207.
Manda, O., Hadjivassiliou, M., Varrassi, G., Zavridis, P., Zis, P. (2025). Exploring the Role of the Cerebellum in Pain Perception: A Narrative Review. Pain and Therapy, 14(3), 803-816. doi: 10.1007/s40122-025-00724-8.
Roychoudhury, S., Buza, N. (2025). Endometriosis Then and Now: A 100-Year Journey Around Pathogenesis and Clinicopathologic Associations. Archives of pathology & laboratory medicine, 150(1), 12-18. doi: 10.5858/arpa.2025-0309-RA.
Warren, A., Garrett, K., Frame, L.A. (2025). Disparities in women’s health and clinical considerations from a translational science perspective: A narrative review and framework for future directions. Women’s Health, 21. doi:10.1177/17455057251399009
Bougie, O., Nwosu, I., Warshafsky, C. (2022). Revisiting the impact of race/ethnicity in endometriosis. Reproduction and Fertility, 3(2), R34–R41. doi: 10.1530/RAF-21-0106
De Corte, P., Klinghardt, M., von Stockum, S. and Heinemann, K. (2025). Time to Diagnose Endometriosis: Current Status, Challenges and Regional Characteristics—A Systematic Literature Review. BJOG: An International Journal of Obstetrics & Gynaecology, 132, 118-130. doi: 10.1111/1471-0528.17973
Ellis, K., Munro, D., Clarke, J. (2022). Endometriosis Is Undervalued: A Call to Action. Frontiers in Global Women's Health, 3. doi: 10.3389/fgwh.2022.902371.