A Literary Classic: Oxytocin and Dopamine in Autism Spectrum Disorder
Sydney Keenan
Illustrations by Abigail Schoenecker
Note: This article uses person-first language to refer to Autism Spectrum Disorder.
From the moment we’re born, social interactions begin to shape our lives [1]. Whether it be hearing our parents’ voices or seeing their facial expressions, these early interactions serve as the foundation for more complex social behaviors, including conversations and relationships [2]. As we develop, we learn how to interpret social cues and respond to others’ emotions [2]. Nevertheless, for some individuals, navigating the social world is not as intuitive as it may be for others. While many children relish in the chaos of their school cafeteria, eager to exchange jokes and stories with their peers, some may find this environment challenging. For individuals with autism spectrum disorder (ASD), interpreting facial expressions or maintaining reciprocal conversations with their peers may be difficult, leading to social interactions that can feel confusing, unpredictable, or overwhelming [3, 4, 5]. ASD is a neurodevelopmental disorder associated with various social deficits, alongside restrictive and repetitive behaviors or fixation of interests [6, 7, 8]. Certain individuals with ASD may seek treatment to improve their quality of life and alleviate the challenges of socializing with others. Therefore, research into the possible mechanisms underlying ASD can guide avenues for therapy development. Nevertheless, for many individuals, ASD is not something to be ‘treated,’ but rather a different way of experiencing the world that must be better understood.
Sense and Sociability: Behavior and Biology
A lifelong social journey begins when a newborn baby catches their parent’s gaze and beams back. Humans are biologically wired to form connections with others [1]. From infancy, we possess specialized cognitive processes for navigating the social world, including an innate ability to detect and recognize faces [1, 9]. As we develop, we expand our repertoire of social skills — such as verbal communication, empathy, cooperation, and conflict resolution — through experience and interactions with others [1]. In turn, these social skills guide two broad categories of social behavior: affiliative and agonistic [10, 11]. Affiliative behaviors, like a child happily sharing their toys with other kids, are interactions between individuals that develop, maintain, and strengthen social bonds [10]. In contrast, agonistic behaviors are often associated with conflict and can encompass actions like a child withdrawing from conversation or avoiding peers [11]. Both types of behaviors begin to manifest as individuals develop social complexity, usually occurring during the early stages of childhood [12]. These trends continue into adulthood, influencing how individuals form romantic partnerships, collaborate with coworkers, and learn conflict resolution [12]. Despite similar patterns in how people interact with one another, sociability can vary greatly from person to person [13]. Some people are more introverted and require periods of solitude to recharge their social battery, while others are more extroverted and can feel recharged by social interactions and group settings [14]. However, in some cases, differences in social behavior may be more prominent and can interfere with an individual’s daily functioning, leading to the classification of a disorder [15, 16, 17]. Understanding these differences can provide critical information for developing personalized therapies for behavioral and cognitive disorders, as well as establishing a stronger understanding of the background of disorders such as ASD [13].
The Odyssey: A Journey into ASD
ASD is characterized by differences in social cognition, repetitive behaviors, fixation of interests, and atypical perception, including heightened sensitivity to sound, light, and texture [6, 7, 8]. Symptoms are typically categorized into two domains: deficits in social communication and interaction, and restricted, repetitive patterns of behavior [6]. A child with autism may struggle to make friends because they have difficulties initiating or maintaining conversation across a loud cafeteria table. Additionally, environments like cafeterias can be overstimulating, with distracting sensory inputs such as the stickiness of the table and the strong smell of lunch meat. Furthermore, many individuals with ASD also prefer a strict and predictable routine, so someone else sitting in their usual spot in the cafeteria can feel overly stressful. Nevertheless, ASD symptoms can vary tremendously person-to-person, which made prior attempts to categorize ASD under a single, universal set of parameters difficult [18, 19]. It wasn’t until 2013 that the concept of autism as a spectrum was first presented, leading to an expansion of diagnostic criteria that considered the variability between the number and severity of ASD symptoms [18, 19]. Subsequently, this broadening of diagnostic criteria has likely contributed to the increased number of ASD diagnoses observed in recent decades [18, 20, 21].
Despite the increased prevalence of ASD in recent years, the mechanisms and possible causes behind the disorder remain relatively unclear [22]. ASD may arise from a combination of genetic and environmental factors, such as gene mutations, maternal infection, higher parental age, and hormonal imbalance that affect the developing brain [18, 23]. As there is still so much to learn about ASD, there are many different elements being explored. One promising area of ASD research focuses on neuromodulators — molecules released by brain cells to transmit signals to other cells — that are believed to be involved in both the onset of ASD and the persistence of symptoms [24 25]. While several mechanisms are being studied in relation to ASD, two neuromodulators in particular have stood out due to their role in social behaviors: dopamine and oxytocin [26].
Great Expectations: Dopamine and Social Behavior
Dopamine, primarily produced in the ventral tegmental area (VTA) — a brain region known for regulating reward processing, learning, and memory — is most commonly known for its role in reinforcing behavior [27, 28, 29]. Neuromodulators like dopamine are released between neurons, brain cells that process and communicate information throughout the brain and body [30]. Dopamine release from the VTA has been theorized to facilitate reinforcement-learning, a process that shapes future behaviors by encouraging or discouraging actions through a reward or punishment system [27, 31]. The process of reinforcement learning is believed to occur via a phenomenon known as ‘reward prediction error,’ which hypothesizes that the activity of dopamine-releasing neurons helps encode the difference between a predicted and received reward [27, 31, 32] When a reward is better than expected, dopamine release increases relative to an individual's baseline, and when a reward is worse than expected, dopamine release drops below their baseline [27, 32]. To incentivize you to do your chores, your parents might decide to reward you with chocolate at dinner instead of vegetables. If you received double the amount of chocolate you were expecting, neurons in the VTA would release more dopamine to encode an outcome that was better than expected. Conversely, if you completed all your chores and hoped to receive chocolate with dinner but your parents still served you vegetables, VTA neurons would release less dopamine than normal to encode that this outcome was worse than expected.
Subsequently, these changes in the firing of VTA neurons can influence behaviors through the process of reinforcement learning by forming new reward associations and updating previously learned associations [27, 33].During the first week, when you do your chores and get twice the chocolate you expected, dopamine release increases, helping your brain form the association and incentivizing you to complete your work in the future. Yet, in the second week, when you don’t receive chocolate despite doing your chores, the relative decrease in dopamine can update your previously formed association, telling your brain that chores don’t always equal chocolate. The updated association can discourage you from completing chores in the future, because the outcome you want is not always guaranteed. A similar pattern occurs in social behavior where a positive reward prediction error, caused by a reward that was better than expected and increased dopamine release from the VTA, often leads to affiliative behaviors [33]. Conversely, a negative reward prediction error, or decreased dopamine release, can contribute to agonistic behaviors [33].Adolescents with ASD may experience dysregulated reward prediction errors in response to social stimuli, but not non-social stimuli [34]. Dysfunction of this ability to assign values to social stimuli likely contributes to atypical social reinforcement learning and decreased motivation to engage in social interactions, commonly seen in individuals with ASD [34, 35].
A Tale of Two Cities: Dopamine and Oxytocin
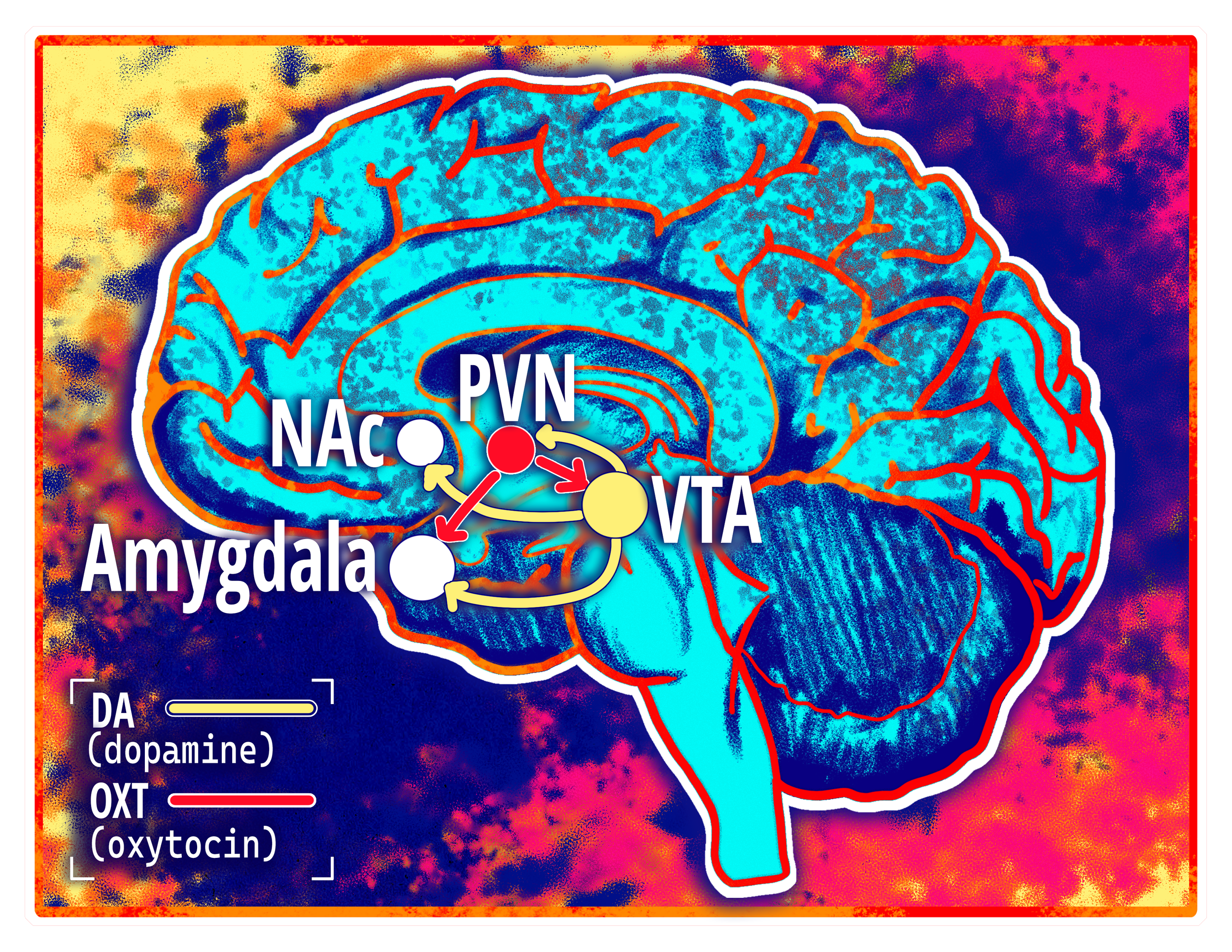
In addition to dopamine, oxytocin plays a key role in many social behaviors, and both neuromodulators regulate one another’s release through multiple mechanisms [36, 37, 38]. Oxytocin influences a multitude of social behaviors, including social recognition and forming close relationships with other individuals [39, 40]. It is primarily produced in the paraventricular nucleus of the hypothalamus (PVN), a regioninvolved in controlling diverse survival behaviors such as appetite, alertness, social and defensive behavior, and pain [41]. The PVN delivers oxytocin to numerous regions of the brain, including the VTA, where it binds onto dopamine neurons [36, 42]. Oxytocin increases dopamine release from the VTA into brain regions such as the nucleus accumbens (NAc), helping the brain encode positive social value via reward prediction error [36, 43, 44]. Therefore, oxytocin can influence how rewarding a behavior ‘feels’ by modulating dopamine release from the VTA to the NAc [36, 45].
Oxytocin release from the PVN also serves a key role in enhancing the salience, or noticeability, of social stimuli, which help guide social behavior [46, 47, 48]. By directing the brain’s focus toward social stimuli instead of background distractions, individuals can focus on signals significant to the social environment [47, 48]. Oxytocin elicits this effect by regulating various sensory pathways, notably through its interaction with the brain’s dopamine system [46, 47, 48]. Similarly to how oxytocin regulates reward-encoding dopamine neurons, oxytocin is also believed to modulate the salience of social stimuli by regulating dopamine firing [46]. Unlike the dopamine neurons involved in reward prediction error, which only increase activity in response to an unexpected positive reward, these dopamine neurons increase firing in response to any salient stimuli, positive or negative [43]. Oxytocin, therefore, functions like a biological DJ turntable by selectively amplifying social signals — such as visual and auditory cues — while dampening non-social signals to help the brain prioritize signals most important to social interaction [49, 50].
Oxytocin and dopamine signaling also regulate social behavior by modulating the activity of the amygdala [51]. The amygdala is a brain region mostly known for its role in processing emotions and regulating fear and anxiety responses, which in turn contribute to various social behaviors [52, 53]. Increased oxytocin release can help encourage social engagement by decreasing anxiety-related signaling from the amygdala [51, 54]. However, it is believed that oxytocin does not elicit these effects on its own; instead, it interacts with other neuromodulators such as dopamine to produce them [51]. Oxytocin and dopamine receptors are found together throughout the amygdala; therefore, it is likely that both oxytocin and dopamine signaling contribute to the regulation of anxiety-related behaviors and, in turn, promote affiliative behaviors [51, 55, 56].
The Neuromodulator’s Tale: The Interplay of Oxytocin and Dopamine
Since oxytocin and dopamine mutually influence one another, dysfunction in both neuromodulators has been implicated in ASD [36]. Dysregulated oxytocin and dopamine signaling can cause atypical social reward processing and social salience encoding [57, 58]. This dysfunction likely contributes to social deficits associated with ASD, like avoiding eye contact or becoming overstimulated by certain environments [36, 59, 60]. Deviations in oxytocin levels can disrupt the regulation of dopamine neurons in the VTA, often resulting in decreased dopamine release [36, 42, 45]. Dopamine signaling between the VTA and NAc helps encode reward and reinforce behavior, and thus, reductions in dopamine release can disrupt social reward processing [27, 33, 43]. Therefore, individuals with ASD may not get as much reinforcement from positive social interactions, causing them to feel less motivated to engage or initiate them in the future [33].
Dysfunction in oxytocin-dopamine signaling has a similar effect on social salience, as dysregulated release can impair the brain's ability to differentiate the importance of social stimuli relative to unrelated ‘background noise’ [46]. As a result, individuals with ASD experience difficulty socializing, as too much sensory input and background noise can make it challenging for the brain to filter and process social cues [37, 38, 61]. In a noisy environment such as a playground, a child with ASD might struggle to notice a friend waving or calling their name, as dysregulated oxytocin and dopamine signaling make it hard for their brain to mark these social cues as important amidst all the other sensory stimuli. Furthermore, this may lead to a feedback loop of feeling left out or having difficulty making new friends. Dysfunction in oxytocin-dopamine signaling can also contribute to emotional challenges associated with ASD, such as atypical regulation of the amygdala [57]. When oxytocin and dopamine bind to receptors on the amygdala, they can help regulate amygdala activity and decrease anxiety-related signals, subsequently promoting affiliative behaviors [51, 55, 56]. When this regulation is disrupted, individuals may exhibit heightened anxiety and reduced motivation to engage in social interactions — core features associated with ASD [57, 62].
Catch-22: ASD and Pharmacological Therapies
Research into ASD all shares the same primary goal: to increase knowledge to improve the quality of life and long-term outcomes for individuals with autism and their families [63, 64]. Interventions such as pharmacological and behavioral therapies do not aim to fix or eliminate autism, but rather strive to manage symptoms [65, 66]. Importantly, therapies for autism are often used in conjunction with one another to holistically address symptoms [67]. Despite oxytocin and dopamine dysfunction in autism still being a relatively new area of research, some clinical trials targeting these neuromodulators have shown promise [68, 69, 70]. In both children and adults with ASD, oxytocin administration in children and adolescents with ASD has been shown to improve various symptoms [68, 71, 72]. Adults with ASD showed a significant reduction in repetitive behaviors for up to four hours after administration of oxytocin and increased attention to social cues, such as others' eyes or mouths [68, 72]. In children and adolescents with ASD, oxytocin has been shown to improve social abilities such as communication, motivation, and awareness [71].
Much like oxytocin, clinical trials testing medications targeting dopamine deficits have also shown efficacy in treating associated ASD symptoms [69, 70]. There are only two medications currently approved by the Food and Drug Administration (FDA) for symptoms associated with ASD: risperidone and aripirazole, both of which target dysregulated dopamine signaling [73, 74]. Children treated with risperidone exhibited decreased sensory processing abnormalities, decreased hypersensitivity, and decreased irritability [69]. Similarly, aripiprazole was shown to improve symptoms associated with ASD, such as irritability, mood swings, and severe tantrums [75]. Though not developed for ASD treatment, clozapine, believed to increase dopamine release, yielded similar results to aripiprazole when taken for management of ASD symptoms [70]. Specifically, clozapine administration was found to decrease severe disruptive behaviors such as self-injury and tantrums [70, 76]. But due to severe side effects including seizures, decreased immune response, and heart issues, it is only used as a last resort intervention for people with severe, treatment-resistant symptoms [70]. Despite encouraging results after treatment, research into the efficacy and safety of these interventions remains limited [77, 78]. Continued research into oxytocin and dopamine dysfunction in ASD is critical for guiding future clinical trials aiming to discover new pharmacological therapies [24, 79, 80].
The Metamorphoses: Advancement in ASD Research
ASD reflects a complex interaction between neurobiology and behavior, particularly in neural systems that regulate social reward, perception, and motivation [81]. Research into the neuromodulators oxytocin and dopamine has begun to clarify how dysfunction in social reward processing and social salience encoding contributes to social challenges experienced by individuals with ASD [62]. While these findings do not insinuate that autism is something that needs to be ‘fixed,’ they offer valuable insight for understanding how individuals with ASD experience and navigate the social world [65, 66, 82]. Rather than seeking out a single cause or universal treatment, ASD research aims to develop a more nuanced and individualized understanding that emphasizes the variability of the disorder and the need for personalized interventions [83, 84]. Of course, the decision to seek treatment is dependent on the individual, and many people with ASD do not wish to seek therapeutic intervention. As research into ASD continues to evolve, it holds promise for developing targeted pharmacological and behavioral interventions that work to alleviate symptoms and improve quality of life — ensuring individuals with ASD have access to tools and support that can help them better navigate social environments [24, 65, 66].
References
Ilyka, D., Johnson, M. H., & Lloyd-Fox, S. (2021). Infant social interactions and brain development: A systematic review. Neuroscience & Biobehavioral Reviews, 130, 448–469. https://doi.org/10.1016/j.neubiorev.2021.09.001
Leitzke, B. T., Cochrane, A., Stein, A. G., DeLap, G. A., Green, C. S., & Pollak, S. D. (2024). Children’s and adolescent’s use of context in judgments of emotion intensity. Affective Science, 6(1), 117–127. https://doi.org/10.1007/s42761-024-00279-5
Keating, C. T., & Cook, J. L. (2021). Facial expression production and recognition in autism spectrum disorders. Psychiatric Clinics of North America, 44(1), 125–139. https://doi.org/10.1016/j.psc.2020.11.010
Kern Koegel, L., Ashbaugh, K., Navab, A., & Koegel, R. L. (2016). Improving empathic communication skills in adults with autism spectrum disorder. Journal of Autism and Developmental Disorders, 46(3), 921–933. https://doi.org/10.1007/s10803-015-2633-0
Boldsen, S. (2022). Autism and the sensory disruption of Social Experience. Frontiers in Psychology, 13. https://doi.org/10.3389/fpsyg.2022.874268
Neurodevelopmental disorders. (2022). Diagnostic and Statistical Manual of Mental Disorders. https://doi.org/10.1176/appi.books.9780890425787.x01_neurodevelopmental_disorders
Frye, R. E. (2018). Social skills deficits in autism spectrum disorder: Potential biological origins and progress in developing therapeutic agents. CNS Drugs, 32(8), 713–734. https://doi.org/10.1007/s40263-018-0556-y
Wang, L., Wang, B., Wu, C., Wang, J., & Sun, M. (2023). Autism spectrum disorder: Neurodevelopmental risk factors, biological mechanism, and precision therapy. International Journal of Molecular Sciences, 24(3), 1819. https://doi.org/10.3390/ijms24031819
Talaat, F. M. (2023). Real-time facial emotion recognition system among children with autism based on Deep Learning and IOT. Neural Computing and Applications, 35(17), 12717–12728. https://doi.org/10.1007/s00521-023-08372-9
Jasso del Toro, C., & Nekaris, K. A.-I. (2022). Affiliative behaviors. Encyclopedia of Animal Cognition and Behavior, 106–111. https://doi.org/10.1007/978-3-319-55065-7_1040
Young, C. (2019). Agonistic behavior. Encyclopedia of Animal Cognition and Behavior, 1–6. https://doi.org/10.1007/978-3-319-47829-6_320-1
Hay, D. F., Paine, A. L., Perra, O., Cook, K. V., Hashmi, S., Robinson, C., Kairis, V., & Slade, R. (2021). Prosocial and aggressive behavior: A longitudinal study. Monographs of the Society for Research in Child Development, 86(2), 7–103. https://doi.org/10.1111/mono.12427
Dickerson, B. C. (2020). Dysfunction of social cognition and behavior. Continuum, 21(3), 660–677. https://doi.org/10.1212/01.con.0000466659.05156.1d
Tuovinen, S., Tang, X., & Salmela-Aro, K. (2020). Introversion and social engagement: Scale validation, their interaction, and positive association with self-esteem. Frontiers in Psychology, 11. https://doi.org/10.3389/fpsyg.2020.590748
Golya, N., & McIntyre, L. L. (2017). Variability in adaptive behaviour in young children with autism spectrum disorder. Journal of Intellectual & Developmental Disability, 43(1), 102–111. https://doi.org/10.3109/13668250.2017.1287886
Hur, J., DeYoung, K. A., Islam, S., Anderson, A. S., Barstead, M. G., & Shackman, A. J. (2019). Social context and the real-world consequences of social anxiety. Psychological Medicine, 50(12), 1989–2000. https://doi.org/10.1017/s0033291719002022
Moraleda Sepúlveda, E., & López Resa, P. (2021). Evaluating quality of life in families with Williams Syndrome patients. Health and Quality of Life Outcomes, 19(1). https://doi.org/10.1186/s12955-021-01704-0
Hodges, H., Fealko, C., & Soares, N. (2020). Autism spectrum disorder: Definition, epidemiology, causes, and clinical evaluation. Translational Pediatrics, 9(S1). https://doi.org/10.21037/tp.2019.09.09
Rosen, N. E., Lord, C., & Volkmar, F. R. (2021). The diagnosis of autism: From kanner to DSM-III to DSM-5 and beyond. Journal of Autism and Developmental Disorders, 51(12), 4253–4270. https://doi.org/10.1007/s10803-021-04904-1
Grosvenor, L. P., Croen, L. A., Lynch, F. L., Marafino, B. J., Maye, M., Penfold, R. B., Simon, G. E., & Ames, J. L. (2024). Autism diagnosis among US children and adults, 2011-2022. JAMA Network Open, 7(10). https://doi.org/10.1001/jamanetworkopen.2024.42218
Andrade, C. (2025). Autism spectrum disorder, 1. The Journal of Clinical Psychiatry, 86(2). https://doi.org/10.4088/jcp.25f15878
Grabrucker, A. M. (Ed.). (2021). Autism spectrum disorders. Exon Publications. https://doi.org/10.36255/exonpublications.autismspectrumdisorders.2021
Wang, M., Zhang, X., Zhong, L., Zeng, L., Li, L., & Yao, P. (2025). Understanding autism: Causes, diagnosis, and advancing therapies. Brain Research Bulletin, 227, 111411. https://doi.org/10.1016/j.brainresbull.2025.111411
Marotta, R., Risoleo, M. C., Messina, G., Parisi, L., Carotenuto, M., Vetri, L., & Roccella, M. (2020). The neurochemistry of autism. Brain Sciences, 10(3), 163. https://doi.org/10.3390/brainsci10030163
Teleanu, R. I., Niculescu, A.-G., Roza, E., Vladâcenco, O., Grumezescu, A. M., & Teleanu, D. M. (2022). Neurotransmitters—key factors in neurological and neurodegenerative disorders of the Central Nervous System. International Journal of Molecular Sciences, 23(11), 5954. https://doi.org/10.3390/ijms23115954
Guidolin, D., Tortorella, C., Cervetto, C., Marcoli, M., Maura, G., & Agnati, L. F. (2025). Interaction between oxytocin and dopamine signaling: Focus on the striatum. International Journal of Molecular Sciences, 26(17), 8711. https://doi.org/10.3390/ijms26178711
Wang, A. R., Groome, A., Taniguchi, L., Eshel, N., & Bentzley, B. S. (2020). The role of dopamine in reward-related behavior: Shining New Light on an old debate. Journal of Neurophysiology, 124(2), 309–311. https://doi.org/10.1152/jn.00323.2020
Padilla-Coreano, N., & Martínez-Rivera, F. J. (2025). How dopamine guides our social world. Pharmacological Reviews, 77(5), 100085. https://doi.org/10.1016/j.pharmr.2025.100085
Cai, J., & Tong, Q. (2022). Anatomy and function of ventral tegmental area glutamate neurons. Frontiers in Neural Circuits, 16. https://doi.org/10.3389/fncir.2022.867053
Azarfar, A., Calcini, N., Huang, C., Zeldenrust, F., & Celikel, T. (2018). Neural coding: A single neuron’s perspective. Neuroscience & Biobehavioral Reviews, 94, 238–247. https://doi.org/10.1016/j.neubiorev.2018.09.007
Palminteri, S. (2025). Human reinforcement learning processes and biases: Computational characterization and possible applications to behavioral public policy. Mind & Society, 24(2), 249–273. https://doi.org/10.1007/s11299-025-00329-w
Schultz, W. (2016). Dopamine reward prediction error coding. Dialogues in Clinical Neuroscience, 18(1), 23–32. https://doi.org/10.31887/dcns.2016.18.1/wschultz
Solié, C., Girard, B., Righetti, B., Tapparel, M., & Bellone, C. (2021). VTA dopamine neuron activity encodes social interaction and promotes reinforcement learning through social prediction error. Nature Neuroscience, 25(1), 86–97. https://doi.org/10.1038/s41593-021-00972-9
Kinard, J. L., Mosner, M. G., Greene, R. K., Addicott, M., Bizzell, J., Petty, C., Cernasov, P., Walsh, E., Eisenlohr‐Moul, T., Carter, R. M., McLamb, M., Hopper, A., Sukhu, R., & Dichter, G. S. (2020). Neural mechanisms of social and nonsocial reward prediction errors in adolescents with autism spectrum disorder. Autism Research, 13(5), 715–728. https://doi.org/10.1002/aur.2273
Schuetze, M., Rohr, C. S., Dewey, D., McCrimmon, A., & Bray, S. (2017). Reinforcement learning in autism spectrum disorder. Frontiers in Psychology, 8. https://doi.org/10.3389/fpsyg.2017.02035
Petersson, M., & Uvnäs-Moberg, K. (2024). Interactions of oxytocin and dopamine—effects on behavior in health and disease. Biomedicines, 12(11), 2440. https://doi.org/10.3390/biomedicines12112440
Orchard, E. R., Dakin, S. C., & van Boxtel, J. J. (2022). Internal noise measures in coarse and fine motion direction discrimination tasks and the correlation with autism traits. Journal of Vision, 22(10), 19. https://doi.org/10.1167/jov.22.10.19
Limon, A., & Corona-Moreno, M. (2025). Hyperconnectivity and disrupted signal-to-noise processing in autism. The Palgrave Encyclopedia of Disability, 1–9. https://doi.org/10.1007/978-3-031-40858-8_179-1
Jones, C., Barrera, I., Brothers, S., Ring, R., & Wahlestedt, C. (2017). Oxytocin and social functioning. Dialogues in Clinical Neuroscience, 19(2), 193–201. https://doi.org/10.31887/dcns.2017.19.2/cjones
Havranek, T., Bacova, Z., & Bakos, J. (2024). Oxytocin, GABA, and dopamine interplay in autism. Endocrine Regulations, 58(1), 105–114. https://doi.org/10.2478/enr-2024-0012
Iremonger, K. J., & Power, E. M. (2025). The paraventricular nucleus of the hypothalamus: A key node in the control of behavioural states. The Journal of Physiology, 603(8), 2231–2243. https://doi.org/10.1113/jp288366
Hou, G., Hao, M., Duan, J., & Han, M.-H. (2024). The formation and function of the VTA dopamine system. International Journal of Molecular Sciences, 25(7), 3875. https://doi.org/10.3390/ijms25073875
Cross, E. A., Borland, J. M., Shaughnessy, E. K., Lee, S. D., Vu, V., Sambor, E. A., Meisel, R. L., Huhman, K. L., & Albers, H. E. (2025). Distinct subcircuits within the mesolimbic dopamine system encode the salience and valence of social stimuli. Psychopharmacology, 242(10), 2219–2232. https://doi.org/10.1007/s00213-025-06793-z
Rappeneau, V., & Castillo Díaz, F. (2024). Convergence of oxytocin and dopamine signalling in neuronal circuits: Insights into the neurobiology of social interactions across species. Neuroscience & Biobehavioral Reviews, 161, 105675. https://doi.org/10.1016/j.neubiorev.2024.105675
Xiao, L., Priest, M. F., & Kozorovitskiy, Y. (2018). Oxytocin functions as a spatiotemporal filter for excitatory synaptic inputs to VTA dopamine neurons. eLife, 7. https://doi.org/10.7554/elife.33892
Shamay-Tsoory, S. G., & Abu-Akel, A. (2016). The social salience hypothesis of oxytocin. Biological Psychiatry, 79(3), 194–202. https://doi.org/10.1016/j.biopsych.2015.07.020
Froemke, R. C., & Young, L. J. (2021). Oxytocin, neural plasticity, and social behavior. Annual Review of Neuroscience, 44(1), 359–381. https://doi.org/10.1146/annurev-neuro-102320-102847
Triana-Del Rio, R., Ranade, S., Guardado, J., LeDoux, J., Klann, E., & Shrestha, P. (2022). The modulation of emotional and social behaviors by oxytocin signaling in limbic network. Frontiers in Molecular Neuroscience, 15. https://doi.org/10.3389/fnmol.2022.1002846
Fineberg, S. K., & Ross, D. A. (2017). Oxytocin and the social brain. Biological Psychiatry, 81(3). https://doi.org/10.1016/j.biopsych.2016.11.004
Sun, Y., Wang, X., Chen, Y., Luan, Z., & Hao, R. (2025). The impact of exogenous oxytocin on visual cortex plasticity across different stages of visual development. Scientific Reports, 15(1). https://doi.org/10.1038/s41598-025-96573-8
de la Mora, M. P., Pérez-Carrera, D., Crespo-Ramírez, M., Tarakanov, A., Fuxe, K., & Borroto-Escuela, D. O. (2016). Signaling in dopamine D2 receptor-oxytocin receptor heterocomplexes and its relevance for the anxiolytic effects of dopamine and oxytocin interactions in the amygdala of the rat. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease, 1862(11), 2075–2085. https://doi.org/10.1016/j.bbadis.2016.07.004
Šimić, G., Tkalčić, M., Vukić, V., Mulc, D., Španić, E., Šagud, M., Olucha-Bordonau, F. E., Vukšić, M., & R. Hof, P. (2021). Understanding emotions: Origins and roles of the amygdala. Biomolecules, 11(6), 823. https://doi.org/10.3390/biom11060823
Meisner, O. C., Dal Monte, O., Fagan, N., Nandy, A. S., & Chang, S. W. (2025). Oxytocin in the amygdala sustains prosocial behavior via state-dependent amygdala–prefrontal modulation. The Journal of Neuroscience, 45(36). https://doi.org/10.1523/jneurosci.2416-24.2025
Nerio-Morales, L. K., Boender, A. J., Young, L. J., Lamprea, M. R., & Smith, A. S. (2024). Limbic oxytocin receptor expression alters molecular signaling and social avoidance behavior in female prairie voles (microtus ochrogaster). Frontiers in Neuroscience, 18. https://doi.org/10.3389/fnins.2024.1409316
An, X., Yu, P., & Chang, G. (2025). Dopamine type II receptors in amygdala along with oxytocin in hypothalamus regulate social behavior in male Mandarin voles. Pharmacology Biochemistry and Behavior, 250, 174002. https://doi.org/10.1016/j.pbb.2025.174002
Hernández-Mondragón, J. C., Hernández-Hernández, D. A., Crespo-Ramírez, M., Prospero-García, O., Rocha-Arrieta, L., Fuxe, K., Borroto-Escuela, D. O., & Perez de la Mora, M. (2023). Evidence for the existence of facilitatory interactions between the dopamine D2 receptor and the oxytocin receptor in the amygdala of the rat. relevance for anxiolytic actions. Frontiers in Pharmacology, 14. https://doi.org/10.3389/fphar.2023.1251922
Gordon, I., Jack, A., Pretzsch, C. M., Vander Wyk, B., Leckman, J. F., Feldman, R., & Pelphrey, K. A. (2016). Intranasal oxytocin enhances connectivity in the neural circuitry supporting social motivation and social perception in children with autism. Scientific Reports, 6(1). https://doi.org/10.1038/srep35054
Kruppa, J. A., Gossen, A., Oberwelland Weiß, E., Kohls, G., Großheinrich, N., Cholemkery, H., Freitag, C. M., Karges, W., Wölfle, E., Sinzig, J., Fink, G. R., Herpertz-Dahlmann, B., Konrad, K., & Schulte-Rüther, M. (2018). Neural modulation of social reinforcement learning by intranasal oxytocin in male adults with high-functioning autism spectrum disorder: A randomized trial. Neuropsychopharmacology, 44(4), 749–756. https://doi.org/10.1038/s41386-018-0258-7
Madipakkam, A. R., Rothkirch, M., Dziobek, I., & Sterzer, P. (2017). Unconscious avoidance of eye contact in autism spectrum disorder. Scientific Reports, 7(1). https://doi.org/10.1038/s41598-017-13945-5
Millington, E. (2024). Sensorily stressed: an exploration of the relationship between anxiety and sensory reactivity in autistic people. University of Glasgow. https://doi.org/https://doi.org/10.5525/gla.thesis.84280
Zhang, J., Eaton, M., Chen, X., Zhao, Y., Kant, S., Deming, B. A., Harish, K., Nguyen, H. P., Shu, Y., Lai, S., Wu, J., Que, Z., Wettschurack, K. W., Zhang, Z., Xiao, T., Halurkar, M. S., Olivero-Acosta, M. I., Yoo, Y.-E., Lanman, N. A., … Yang, Y. (2025). Restoration of excitation/inhibition balance enhances neuronal signal-to-noise ratio and rescues social deficits in autism-associated scn2a -deficiency. bioRxiv. https://doi.org/10.1101/2025.03.04.641498
Mayer, A. V., Preckel, K., Ihle, K., Piecha, F. A., Junghanns, K., Reiche, S., Rademacher, L., Müller-Pinzler, L., Stolz, D. S., Kamp-Becker, I., Stroth, S., Roepke, S., Küpper, C., Engert, V., Singer, T., Kanske, P., Paulus, F. M., & Krach, S. (2022). Assessment of reward-related brain function after a single dose of oxytocin in autism: A randomized controlled trial. Biological Psychiatry Global Open Science, 2(2), 136–146. https://doi.org/10.1016/j.bpsgos.2021.10.004
Pfeiffer, B., Piller, A., Giazzoni-Fialko, T., & Chainani, A. (2016). Meaningful outcomes for enhancing quality of life for individuals with autism spectrum disorder. Journal of Intellectual & Developmental Disability, 42(1), 90–100. https://doi.org/10.3109/13668250.2016.1197893
Dai, Y., Chen, M., Deng, T., Huang, B., Ji, Y., Feng, Y., Liu, S., & Zhang, L. (2023). The importance of parenting self‐efficacy and social support for family quality of life in children newly diagnosed with autism spectrum disorder: A one‐year follow‐up study. Autism Research, 17(1), 148–161. https://doi.org/10.1002/aur.3061
Maniram, J., Karrim, S. B., Oosthuizen, F., & Wiafe, E. (2022). Pharmacological management of core symptoms and comorbidities of autism spectrum disorder in children and adolescents: A systematic review. Neuropsychiatric Disease and Treatment, Volume 18, 1629–1644. https://doi.org/10.2147/ndt.s371013
Abbeduto, L., & Sahin, M. (2021). Developing and evaluating treatments for the challenges of autism and related neurodevelopmental disabilities. Journal of Neurodevelopmental Disorders, 13(1). https://doi.org/10.1186/s11689-021-09404-y
Alsayouf, H. A., Talo, H., Biddappa, M. L., Qasaymeh, M., Qasem, S., & De Los Reyes, E. (2020). Pharmacological intervention in children with autism spectrum disorder with standard supportive therapies significantly improves core signs and symptoms: A single-center, retrospective case series. Neuropsychiatric Disease and Treatment, Volume 16, 2779–2794. https://doi.org/10.2147/ndt.s277294
Griffiths, J. L., Mishaal, R. A., Nabetani, M., & Goldman, R. D. (2022). Oxytocin for the treatment of autism spectrum disorder in children. Canadian Family Physician, 68(2), 103–105. https://doi.org/10.46747/cfp.6802103
Panda, P. K., Sharawat, I. K., Gupta, D., Palayullakandi, A., Sopanam, S., & Saha, S. (2025). Efficacy and safety of risperidone and aripiprazole in reducing severity of irritability in children with autism spectrum disorder: A randomized controlled trial. Brain and Development, 47(5), 104454. https://doi.org/10.1016/j.braindev.2025.104454
da Rosa, A. L., Bezerra, O. S., Rohde, L. A., & Graeff-Martins, A. S. (2024). Exploring clozapine use in severe psychiatric symptoms associated with autism spectrum disorder: A scoping review. Journal of Psychopharmacology, 38(4), 324–343. https://doi.org/10.1177/02698811241241384
Parker, K. J., Oztan, O., Libove, R. A., Sumiyoshi, R. D., Jackson, L. P., Karhson, D. S., Summers, J. E., Hinman, K. E., Motonaga, K. S., Phillips, J. M., Carson, D. S., Garner, J. P., & Hardan, A. Y. (2017). Intranasal oxytocin treatment for social deficits and biomarkers of response in children with autism. Proceedings of the National Academy of Sciences, 114(30), 8119–8124. https://doi.org/10.1073/pnas.1705521114
Yamasue, H., Okada, T., Munesue, T., Kuroda, M., Fujioka, T., Uno, Y., Matsumoto, K., Kuwabara, H., Mori, D., Okamoto, Y., Yoshimura, Y., Kawakubo, Y., Arioka, Y., Kojima, M., Yuhi, T., Owada, K., Yassin, W., Kushima, I., Benner, S., … Kosaka, H. (2018). Effect of intranasal oxytocin on the core social symptoms of autism spectrum disorder: A randomized clinical trial. Molecular Psychiatry, 25(8), 1849–1858. https://doi.org/10.1038/s41380-018-0097-2
Davico, C., Secci, I., Vendrametto, V., & Vitiello, B. (2023). Pharmacological treatments in autism spectrum disorder: a narrative review. Journal of Psychopathology, 29(1/2). https://doi.org/https://doi.org/10.36148/2284-0249-N251
Osuna-Luque, J., Rodríguez-Ramos, Á., Gámez-del-Estal, M. del, & Ruiz-Rubio, M. (2018). Behavioral mechanisms that depend on dopamine and serotonin in caenorhabditis elegans interact with the antipsychotics risperidone and Aripiprazole. Journal of Experimental Neuroscience, 12. https://doi.org/10.1177/1179069518798628
Bartram, L. A., Lozano, J., & Coury, D. L. (2019). Aripiprazole for treating irritability associated with autism spectrum disorders. Expert Opinion on Pharmacotherapy, 20(12), 1421–1427. https://doi.org/10.1080/14656566.2019.1626825
da Rosa, A. L., da Costa, M. R., Sorato, G. B., Manjabosco, F. de, de Bem, É. B., Dellazari, L., Falcão, A. B., Cia, L. de, Bezerra, O. S., Borges, R. B., Rohde, L. A., & Graeff-Martins, A. S. (2025). Clozapine for treatment-resistant disruptive behaviors in youths with autism spectrum disorder aged 10-17 years: Protocol for an open-label trial. JMIR Research Protocols, 14. https://doi.org/10.2196/58031
Huang, Y., Huang, X., Ebstein, R. P., & Yu, R. (2021). Intranasal oxytocin in the treatment of autism spectrum disorders: A multilevel meta-analysis. Neuroscience & Biobehavioral Reviews, 122, 18–27. https://doi.org/10.1016/j.neubiorev.2020.12.028
Choi, H., Kim, J. H., Yang, H. S., Kim, J. Y., Cortese, S., Smith, L., Koyanagi, A., Dragioti, E., Radua, J., Fusar-Poli, P., Shin, J. I., Cheon, K.-A., & Solmi, M. (2024). Pharmacological and non-pharmacological interventions for irritability in autism spectrum disorder: A systematic review and meta-analysis with the grade assessment. Molecular Autism, 15(1). https://doi.org/10.1186/s13229-024-00585-6
Gora, C., Dudas, A., Vaugrente, O., Drobecq, L., Pecnard, E., Lefort, G., & Pellissier, L. P. (2024). Deciphering autism heterogeneity: A molecular stratification approach in four mouse models. Translational Psychiatry, 14(1). https://doi.org/10.1038/s41398-024-03113-5
Annamneedi, A., Gora, C., Dudas, A., Leray, X., Bozon, V., Crépieux, P., & Pellissier, L. P. (2023). Towards the convergent therapeutic potential of G protein‐coupled receptors in autism spectrum disorders. British Journal of Pharmacology, 182(14), 3044–3067. https://doi.org/10.1111/bph.16216
Lawrence, K. E., Hernandez, L. M., Eilbott, J., Jack, A., Aylward, E., Gaab, N., Van Horn, J. D., Bernier, R. A., Geschwind, D. H., McPartland, J. C., Nelson, C. A., Webb, S. J., Pelphrey, K. A., Bookheimer, S. Y., Dapretto, M., Aylward, E., Bernier, R. A., Bookheimer, S. Y., Dapretto, M., … Wolf, J. (2020). Neural responsivity to social rewards in autistic female youth. Translational Psychiatry, 10(1). https://doi.org/10.1038/s41398-020-0824-8
Kimber, L., Verrier, D., & Connolly, S. (2024). Autistic people’s experience of empathy and the autistic empathy deficit narrative. Autism in Adulthood, 6(3), 321–330. https://doi.org/10.1089/aut.2023.0001
Litman, A., Sauerwald, N., Green Snyder, L., Foss-Feig, J., Park, C. Y., Hao, Y., Dinstein, I., Theesfeld, C. L., & Troyanskaya, O. G. (2025). Decomposition of phenotypic heterogeneity in autism reveals underlying genetic programs. Nature Genetics, 57(7), 1611–1619. https://doi.org/10.1038/s41588-025-02224-z
Manzo, J., Hernández-Aguilar, M. E., Toledo-Cárdenas, M. R., Herrera-Covarrubias, D., Coria-Avila, G. A., Libreros-Jiménez, H. M., Fernández-Cañedo, L., & Ortega-Pineda, L. A. (2025). The long and winding road to understanding autism. NeuroSci, 6(3), 84. https://doi.org/10.3390/neurosci6030084