Drinks on me: How Alcohol Consumption Impacts Your Brain
Daniel Bader
Illustrations by Ruimei Chau and Alexandra Adsit
After barely scraping through his Econ midterm, Joe decides to go out with his friends to distract himself by getting wildly drunk. At the frat house, he is immediately enveloped by the loud music, bright neon lights, and throngs of people dancing. His buddy Brock hands him a shot, quickly followed by a cold beer. A couple of drinks in, Joe is relaxed and has completely forgotten about how he mixed up the concepts of profit and revenue. He quickly loses count of his drinks and fades away, only to get up the next morning feeling sick and unable to remember any details from his epic night. Although Joe wakes up hungover, he successfully achieved what he wanted. As it did for Joe, alcohol intoxication affects the way we perceive the world around us [1, 2]. Alcohol is a psychoactive drug that alters perception, cognition, behavior, and consciousness [3]. Between the potential short-term consequences of intoxication, like interpersonal violence or car accidents, and the long-term effects of chronic consumption, like liver and cardiovascular disease, alcohol is responsible for over 3.3 million deaths annually worldwide [1]. Still, over 40% of the global population consumed alcohol in the last year, since drinking carries tremendous cultural and social value [4]. Alcohol is frequently consumed at parties or used as a social tool, as it facilitates socialization by ‘taking the edge off’ [5]. However, drinking in excess can cause immediate health emergencies and predispose individuals to addiction later in life [5, 6, 7]. Since alcohol is such a common, yet potentially dangerous, element of our society, it is important to understand how drinking alters your physical and mental state if you do choose to indulge [3].
Bottoms Up! Alcohol's Immediate Effect on the Brain
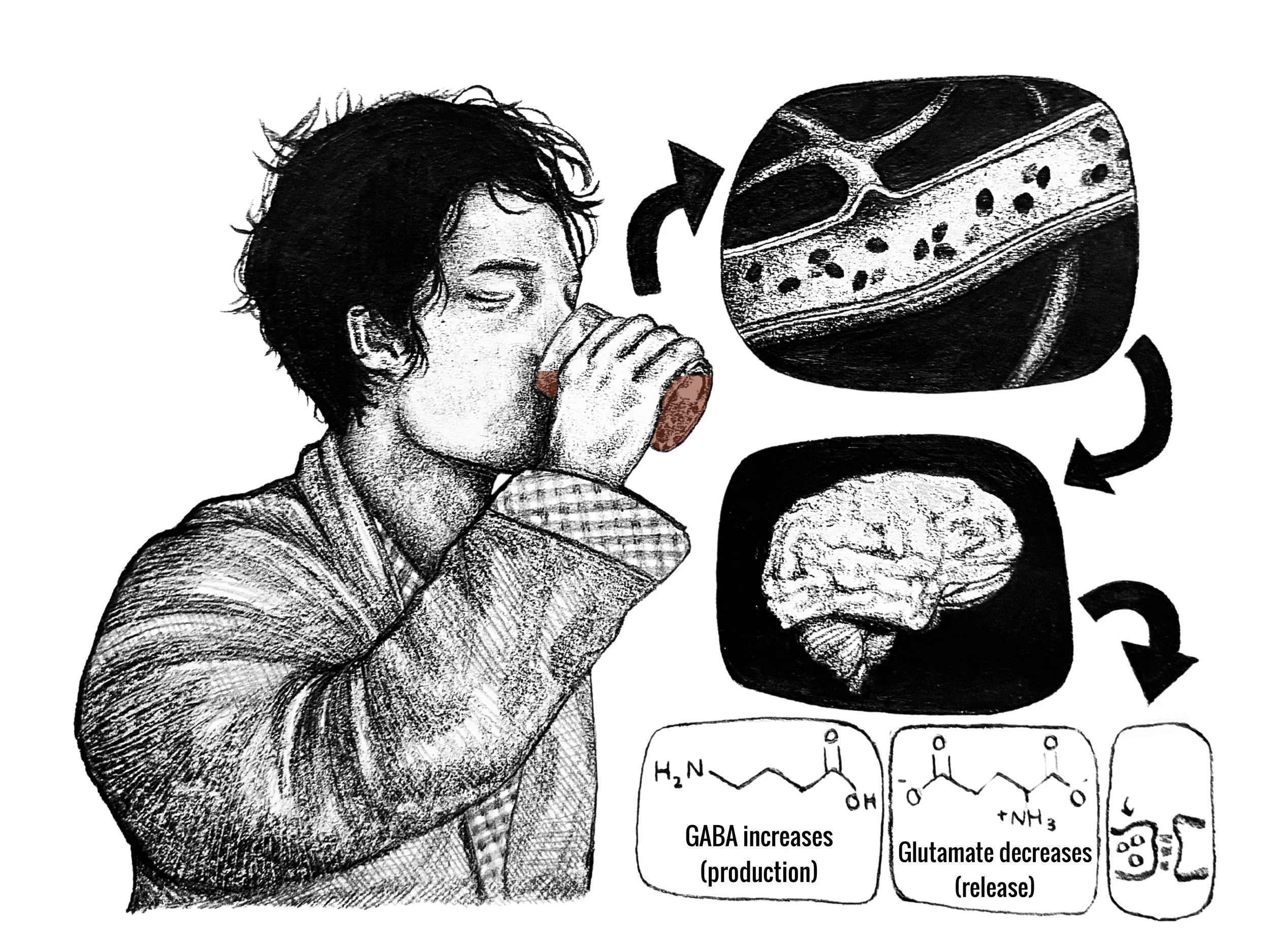
Alcohol is a depressant, meaning that each drink Joe has reduces communication between neurons, cells in the nervous system that process and transmit information [1, 8]. Neural communication is the result of a series of electrical and chemical signals between neurons that relay information in the brain [1, 2, 9]. Signaling is moderated by chemical messengers, called neurotransmitters, which are released by neurons into the space between two neurons, called the synapse [1, 10]. Once neurotransmitters are released into the synapse, they are either absorbed by the adjacent neuron, enabling neuron-to-neuron communication, or broken down and removed [1]. Alcohol significantly changes the concentrations of the two main neurotransmitters that modulate brain activity: GABA and glutamate [1, 4, 9]. GABA is the body’s primary inhibitory neurotransmitter, meaning that its release prevents neural signaling [1, 4]. Conversely, glutamate is an excitatory neurotransmitter that induces neural signaling. As Joe downs his shot, the alcohol enters the bloodstream and flows to the brain, causing an increase in GABA production and a decrease in glutamate release [1, 4]. The combination of increased GABA and decreased glutamate slows neuronal communication, preventing specific brain regions from properly performing their functions [1, 11]. Compare the rate of neuronal communication to the speed of a car: if GABA is the brake pedal and glutamate is the gas pedal, then drinking alcohol pushes the brake while releasing the gas, effectively slowing the car down [1, 4].
Reduced communication within and between brain regions negatively affects motor control and memory until alcohol is fully processed and removed from the body [11, 12]. As Joe goes to get his next drink, he staggers towards his friends, stumbling over his own feet as he dramatically tries to stop himself from falling over. These changes in Joe’s movement and coordination are due to impaired neuronal signalling in the cerebellum, a brain region that is responsible for balance and motor coordination [13, 14]. Similarly, excessive drinking can decrease communication within the hippocampus, a different brain region that manages learning and memory [11]. When Joe got carried away drinking heavily with his friends and woke up the next morning with significant gaps in his memory, he experienced a decrease in hippocampal communication that may lead to the phenomenon colloquially known as ‘blacking out’ [11, 15]. Thankfully for Joe, GABA and glutamate levels return to baseline as alcohol leaves the body, and full motor and memory functionality will return as Joe sobers up [1, 16].
Beyond affecting motor and memory control, intoxication also impacts the brain’s reward system, a group of structures that reinforce behaviors using a neurotransmitter called dopamine [17, 19]. As the last of Joe's sixth beer trickles down his throat, the alcohol increases dopamine secretion into the synapse [1, 17, 18]. Alcohol reduces the efficacy of synaptic proteins that degrade neurotransmitters, leading to unregulated dopamine concentration in the synapse [1]. The prolonged presence of dopamine in the synapse amplifies the pleasure often felt when drinking [1, 17, 18. Any associations Joe makes between drinking and positive feelings can incentivize future drinking behavior [1, 20]. Positive sensations brought on by drinking may outweigh the negative experience that Joe has when he learns about all the embarrassing things he did while intoxicated last Saturday.
In the Eye of the Beer-holder: Individual Differences in Alcohol Tolerance
Hours after his last drink, Joe starts to sober up, but not until his body has cleared most of the alcohol from his system. Although variable, it generally takes the body about one hour to metabolize a standard drink, such as a single can of beer or glass of wine, so that all the alcohol in the bloodstream is degraded [21]. Alcohol clearance is the rate at which alcohol is removed from the bloodstream — a process directly associated with an individual’s alcohol tolerance, or how much a person can drink before becoming intoxicated [21, 22]. Proteins called alcohol dehydrogenases (ADHs) initially break down alcohol into acetaldehyde, a molecule that is toxic at high concentrations and often causes nausea, vomiting, and a rapid pulse [23, 24]. The pounding headache, fatigue, and vertigo ruining Joe’s Sunday morning are likely due to residual acetaldehyde driving the severity of his hangover [25]. Fortunately, hangovers do not last forever. Acetaldehyde is further metabolized into harmless acids by another group of molecules called aldehyde dehydrogenases (ALDH) [24, 26]. Once acetaldehyde is fully broken down and cleared from the bloodstream, most hangover symptoms subside [25].
The amount of time it takes for a person to metabolize alcohol depends on their Blood Alcohol Content (BAC), or the proportion of total body water that is composed of alcohol during periods of drinking [21]. Body water is composed of body fluids and liquids found in tissues, constituting around 50–60% of total body mass. The consumption of any kind of liquid — including your favorite juice, soda, coffee, tea, or 21+ drink — becomes part of the body’s total water content. Joe, who is smaller than his massive, bodybuilder friend Brock, knows that Brock can drink significantly more than he does on nights out without feeling as drunk. Heavier people tend to have more body water than lighter individuals, so long as most of their body weight consists of body fluids and water-dense tissues rather than fat. Imagine adding a single drop of red dye to a small glass of water; the liquid would immediately turn bright red. However, adding a single drop of dye to a large bucket of water would have a more subtle effect. Similarly, since muscle contains more water than body fat, leaner individuals will have a lower BAC after the same number of drinks because they have more relative body water than people of the same weight with a higher body fat percentage. The same logic explains why men typically have higher alcohol tolerance than women, as the latter tend to have higher body fat percentages than men of the same weight [21]. But why can Joe’s smaller friend, Lola, who drinks three times per week, every week, outdrink him every Saturday night? Repeated exposure can also impact alcohol tolerance, as clearance rate increases with habitual drinking, and self-perceptions of intoxication also decrease with subsequent use [27].
Hitting the Bottle Again: Long-Term Effects of Alcohol Abuse on the Immune System
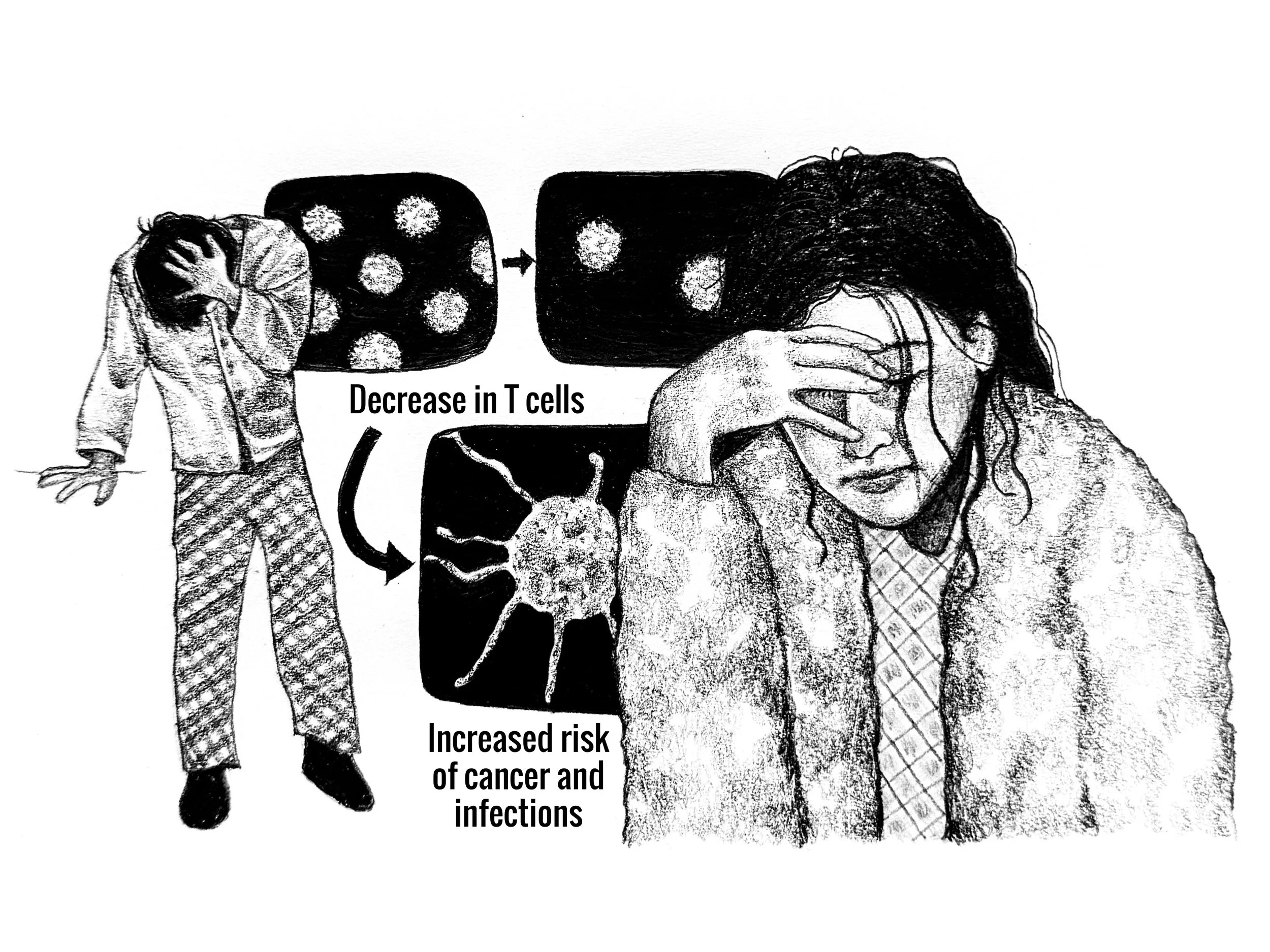
While the short-term effects of intoxication fade away as individuals return to sobriety, alcohol use disorder (AUD) can have long-lasting negative impacts, including decreasing the body’s ability to fight off diseases [28]. AUD is characterized by loss of control over alcohol intake, compulsive alcohol use, and a negative emotional state when not drinking [29, 30]. Alcohol abuse can negatively affect the immune system's ability to fight off pathogens, which are microorganisms that cause disease within a host [28, 31]. The immune system is composed of a complex network of specialized cells, tissues, and organs that defend the body against infection [32]. T cells are specialized cells within the immune system that recognize and eliminate pathogens, recruit other immune cells, and generate long-term disease recognition [32, 33]. At first, T cells are naïve, or unspecialized, meaning they have not yet been activated to respond to a specific pathogen [33, 34]. Some activate to fight infection, and the rest develop to specialize in memory, which supports the immune system in responding more effectively to pathogens it has previously encountered [33, 34]. Unfortunately, AUD reduces the number of naïve T cells, which may increase the risk of developing cancer and infection by limiting the body’s amount of available adaptive cells [34]. Chronic alcohol exposure also acts as a harmful toxin to DNA at the site of T cell maturation, significantly decreasing T cell production and leading to a deficiency [34]. Additionally, alcohol depletes natural killer cells (NKs), a part of the immune system that complements T cells by providing rapid surveillance against a wide range of threats, including virally infected cells and tumors [28, 35]. Therefore, decreases in NKs may explain why AUD is connected to increased susceptibility to cancer of the liver, breast, mouth, pancreas, and colon [28, 36]. Chronic alcohol consumption depletes key immune components, raising the likelihood of infection and disease [28, 34].
While AUD can weaken the immune system and increase vulnerability to pathogens, drinking itself causes further harm since consumption of alcohol directly triggers an immune reaction [37]. In the brain, alcohol activates immune proteins called toll-like receptors (TLRs) [37]. Activation of TLRs triggers the production of proinflammatory cytokines, another class of proteins that generate inflammation at sites of injury and infection [37, 38, 39]. Inflammation plays a crucial role in the immune response by recruiting immune system components to resolve damage, often manifesting as heat and swelling in the affected area [37, 38, 39]. However, prolonged inflammation in the brain, known as neuroinflammation, can cause inflammatory immune cells to target healthy neurons [37, 38]. Picture neuroinflammation as a fire alarm. Stimulated TLRs create commotion and signal for help [40]. Cytokines are like firefighters: they are necessary for putting out fires, but if constantly and unnecessarily involved, they can cause excessive water damage and even more destruction than the fire itself. Hence, while TLRs and the subsequent cytokine response are crucial for immune function, habitual drinking can cause uncontrolled immune-generated neuroinflammation, leading to sustained neuronal toxicity and eventual death [37, 41]. In addition to AUD, chronic TLR activation and subsequent neuroinflammation are hallmarks of many other diseases, including Alzheimer’s disease and Parkinson’s disease [37]. Alzheimer’s is characterized by cognitive and memory impairment, and Parkinson’s often manifests as tremors and walking abnormalities [37, 42]. While these diseases are very different from each other and AUD, they demonstrate how chronic TLR-mediated immune activation can lead to extremely serious neurological complications [37, 42].
Seeing Double: The Two-fold Effect of Alcohol on White and Gray Matter
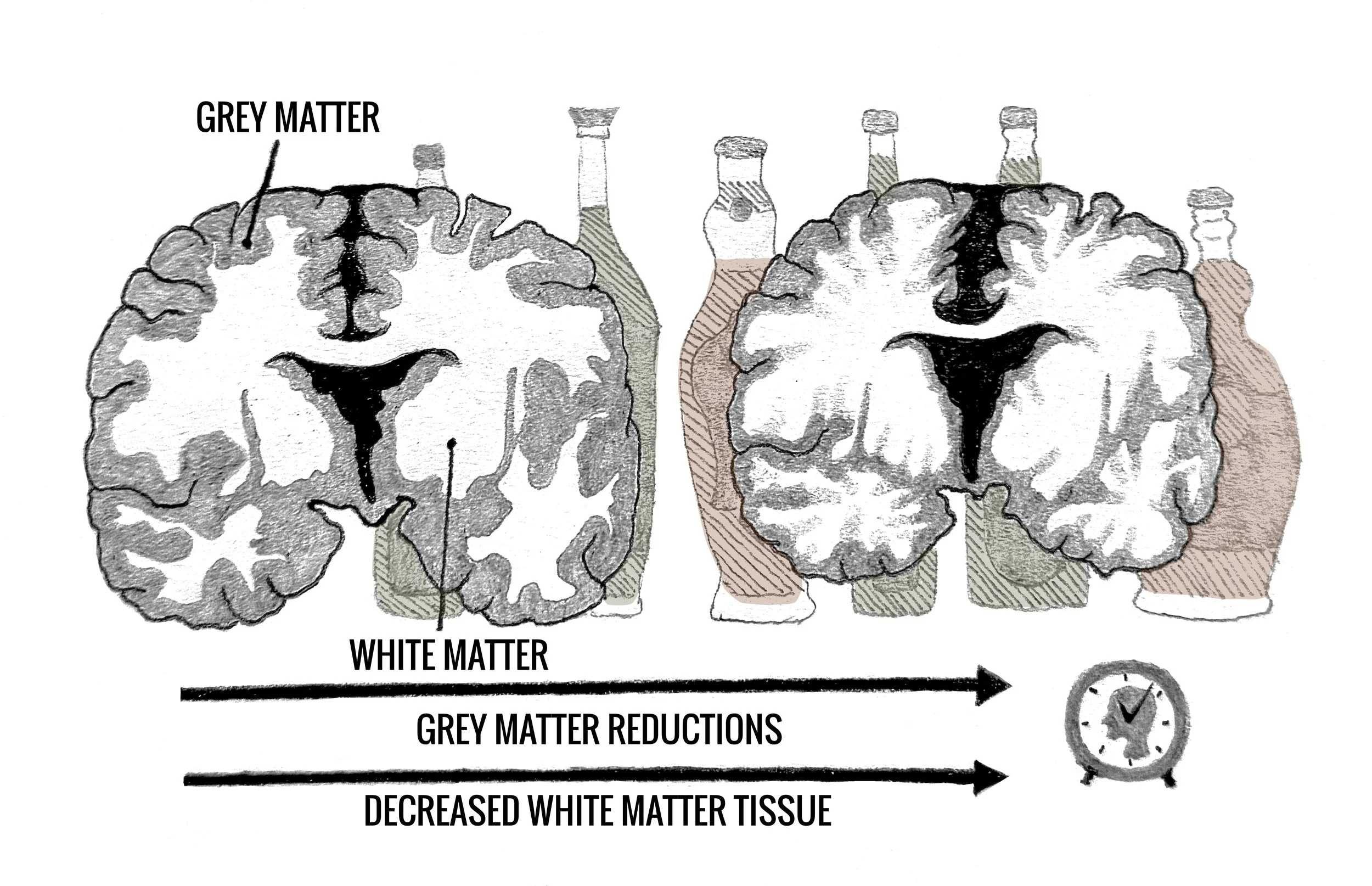
Beyond long-term immune consequences, chronic alcohol abuse can lead to similarly persistent effects on brain structure [43]. One major consequence is atrophy, or shrinking, of brain tissue, a gradual structural change usually associated with aging [43]. Gray matter is the brain tissue that makes up the outer layer of the brain and spinal cord, processing signals between adjacent brain regions [44]. Typical brain aging involves the gradual loss of gray matter volume, which is associated with decreased cognitive efficacy [43, 45]. However, habitual drinkers often experience dramatic gray matter reductions at a rate much faster than natural brain aging [43]. The brain age paradigm, which uses images and scans of brain structures to predict the age of tested individuals, has found that alcohol abuse structurally ages the brain past its biological age by several years, in some cases by over a decade [46, 47]. In essence, habitual drinking acts like a fast-forward button for the brain’s biological clock [48]. Alcohol-induced gray matter reductions are commonly seen in the brain structure primarily responsible for impulse control, called the anterior cingulate cortex (ACC) [49, 50, 51]. These changes in the ACC may lead to impulsivity and failure to consider long-term consequences, such as driving home after drinking despite altered reflexes and judgment [52]. Additionally, alcohol abuse can cause gray matter volume in the hippocampus to shrink, leading to long-term learning and memory issues [53]. AUD specifically impairs recollection of episodic memory, which includes the conscious recollection of specific personal events from the past and the imagination of anticipated events in the future. These impairments occur because alcohol abuse decreases individuals’ ability to organize events chronologically and remember the spatial context of memory, like the layout of the room or who was next to them when watching the finale of their favorite show [53].
Similar to AUD-induced gray matter reductions, drinking is associated with decreases in white matter, which can negatively affect one’s ability to think critically and solve complex problems [54, 55]. White matter is tissue primarily composed of axons, the components of neurons that span long distances in the brain and send signals to the rest of the nervous system [54, 56]. White matter axons can transfer information between hemispheres, a process necessary to ensure executive functioning, a set of cognitive skills that allows individuals to work through novel challenges and maintain focus, such as when solving a puzzle or building a Lego set [57, 58]. Natural aging typically involves a slight decrease in white matter volume, leading to certain cognitive deficits [54]. Sharper rates of deterioration are indicative of memory disorders like dementia, characterized by a decline in cognitive functioning that interferes with daily life without compromising consciousness or perception [54, 59]. While the volume of gray and white matter has been shown to partially recover with abstinence from alcohol, prolonged drinking can cause permanent structural changes that may have lasting detrimental effects on a person's physical and mental health [48].
When it Rains, it Pours: The Cascading Effect of Alcohol on Brain Health
Structural changes in the brain as a result of alcohol abuse can predispose individuals to a variety of neurological disorders [59, 60]. Excessive drinking is connected to cognitive decline, and alcohol abuse doubles the risk of dementia in elderly populations [59]. Cognitive decline in AUD populations may manifest as issues with sensory processing, memory, social cognition, walking, and balance. Although underlying structural causes are distinct, AUD-induced dementia can often mimic the symptomology of Alzheimer’s disease [59]. Individuals with AUD can also develop dementia as a by-product of other AUD-induced diseases [1, 60]. For example, Marchiafava-Bignami disease (MBD) is a neurological disease common in men over 45 with a history of alcohol abuse [1, 60]. MBD is caused by neuronal damage and cell death in the corpus callosum, a large white matter tract that connects the two hemispheres of the brain [1, 60, 61]. As the connection between brain hemispheres is impeded, individuals may experience issues with muscle control, mood disorders, or dementia, and may even enter comatose states [1, 60]. Thus, AUD-induced dementia can also manifest through the development of other neurological diseases [1].
Additionally, AUD can cause nutritional deficiencies that may lead to further devastating neurological conditions [1]. People suffering from alcohol abuse often get a significant part of their caloric intake from nutritionally deficient alcoholic beverages, which may lead to shortages in essential dietary vitamins like thiamine (Vitamin B1) [1, 62]. Thiamine is necessary for nerve function, so a shortage of this crucial vitamin can result in neuron damage [62, 63]. AUD can also contribute to thiamine deficiency since habitual drinking increases stomach acidity, which harms mucous layers in the gut [48]. Pumps in the mucous layer that transport thiamine into the bloodstream require a specific pH level to function and are ineffective with increased acidity [64]. Eventually, untreated thiamine deficiency can develop into Wernicke encephalopathy (WE) — a neurological disease characterized by extreme confusion, visual disturbances, and poor muscular control [1]. WE is reversible, but if left untreated, it can lead to permanent memory and behavioral impairments [65]. Hence, chronic drinking acts as a catalyst for severe and often irreversible neurodegenerative conditions [1, 65].
To Drink, or Not to Drink?
While drinking has been and will remain an important cultural and societal practice, it is important to recognize the potential risk that alcohol poses toward brain health [6]. To practice safety when drinking, it is crucial to understand how inebriation immediately impacts the brain; by altering neurotransmission, intoxication innately leads to memory and cognitive impairments, physical changes, and impulsivity [11, 52]. While the altered mental and physical state brought on by drinking may be sought after in social scenarios, it can be extremely dangerous in the wrong setting, like behind the wheel of a car or in the deep end of a swimming pool. Furthermore, as drinking is popularly practiced across cultures, continents, and demographics, it is important to navigate potential risk factors for developing AUD, like drinking during adolescence [6, 66]. Additionally, alcohol abuse can both lead to depressive or anxious symptoms, while also possibly being misused as an emotional crutch for those already suffering from depression or anxiety [67]. Learning to tread the fine line is important; drinking can be fun and sometimes helpful in social settings, but the innately addictive nature of alcohol combined with the catastrophic neurological effects of AUD cannot be overstated [17, 59].
References
Sahu, P., Verma, H. K., & Bhaskar, L. (2025). Alcohol and alcoholism associated neurological disorders: Current updates in a global perspective and recent recommendations. World Journal of Experimental Medicine, 15(1).doi: 10.5493/wjem.v15.i1.100402
Garrisson, H., Scholey, A., Ogden, E., & Benson, S. (2021). The effects of alcohol intoxication on cognitive functions critical for driving: A systematic review. Accident Analysis & Prevention, 154..doi:10.1016/j.aap.2021.106052
Hamed, M. A., Ekundina, V. O., & Akhigbe, R. E. (2023). Psychoactive drugs and male fertility: Impacts and mechanisms. Reproductive Biology and Endocrinology, 21(1), 69.doi:10.1186/s12958-023-01098-2
Wiegmann, C., Mick, I., Brandl, E. J., Heinz, A., & Gutwinski, S. (2020). Alcohol and Dementia – What is the Link? A Systematic Review. Neuropsychiatric Disease and Treatment, 16, 87–99.doi:10.2147/NDT.S198772
Chung, T., Creswell, K. G., Bachrach, R., Clark, D. B., & Martin, C. S. (2018). Adolescent Binge Drinking. Alcohol Research : Current Reviews, 39(1), 5–15.doi:10.35946/arcr.v39.1.02
Sudhinaraset, M., Wigglesworth, C., & Takeuchi, D. T. (2016). Social and Cultural Contexts of Alcohol Use. Alcohol Research : Current Reviews, 38(1), 35–45.doi:10.35946/arcr.v38.1.05
Orio, L., Alen, F., Pavón, F. J., Serrano, A., & García-Bueno, B. (2019). Oleoylethanolamide, Neuroinflammation, and Alcohol Abuse. Frontiers in Molecular Neuroscience, 11.doi:10.3389/fnmol.2018.00490
Azarfar, A., Calcini, N., Huang, C., Zeldenrust, F., & Celikel, T. (2018). Neural coding: A single neuron’s perspective. Neuroscience & Biobehavioral Reviews, 94, 238–247.doi:10.1016/j.neubiorev.2018.09.007
Wu, Z., Lin, D., & Li, Y. (2022). Pushing the frontiers: Tools for monitoring neurotransmitters and neuromodulators. Nature Reviews Neuroscience, 23(5), 257–274.doi:10.1038/s41583-022-00577-6
Burkhardt, P., & Sprecher, S. G. (2017). Evolutionary origin of synapses and neurons – Bridging the gap. BioEssays, 39(10).doi:10.1002/bies.201700024
Harrison, N. L., Skelly, M. J., Grosserode, E. K., Lowes, D. C., Zeric, T., Phister, S., & Salling, M. C. (2017). Effects of acute alcohol on excitability in the CNS. Neuropharmacology, Alcoholism, 122, 36–45.doi:10.1016/j.neuropharm.2017.04.007
Maestú, F., de Haan, W., Busche, M. A., & DeFelipe, J. (2021). Neuronal excitation/inhibition imbalance: Core element of a translational perspective on Alzheimer's pathophysiology. Ageing Research Reviews, 69.doi:10.1016/j.arr.2021.101372
De la Monte, S. M., & Tong, M. (2025). Molecular and biochemical pathologies in human alcohol-related cerebellar white matter degeneration. Advances in Drug and Alcohol Research, 5.doi:10.3389/adar.2025.15342
Schmahmann, J. D. (2019). The cerebellum and cognition. Neuroscience Letters, The Cerebellum in Health and Disease, 688, 62–75.doi:10.1016/j.neulet.2018.07.005
Hermens, D. F., & Lagopoulos, J. (2018). Binge Drinking and the Young Brain: A Mini Review of the Neurobiological Underpinnings of Alcohol-Induced Blackout. Frontiers in Psychology, 9. doi:10.3389/fpsyg.2018.00012
Urbanik, A., Kozub, J., Karcz, P., & Ostrogórska, M. (2021). Changes in the brain directly following alcohol consumption—A study of healthy male individuals, with the use of proton magnetic resonance spectroscopy (1HMRS) and diffusion (DWI). Alcohol and Alcoholism, 56(4), 415–424.doi:10.1093/alcalc/agaa119
Dahchour, A., & Ward, R. J. (2022). Changes in Brain Dopamine Extracellular Concentration after Ethanol Administration; Rat Microdialysis Studies. Alcohol and Alcoholism, 57(2), 165–175.doi:10.1093/alcalc/agab072
Chau, P., Lidö, H. H., Söderpalm, B., & Ericson, M. (2018). Acamprosate’s ethanol intake-reducing effect is associated with its ability to increase dopamine. Pharmacology Biochemistry and Behavior, 175, 101–107.doi:10.1016/j.pbb.2018.09.009
Costa, K. M., & Schoenbaum, G. (2022). Dopamine. Current Biology, 32(15), 817–824.doi:10.1016/j.cub.2022.06.060
Telzer, E. H. (2016). Dopaminergic reward sensitivity can promote adolescent health: A new perspective on the mechanism of ventral striatum activation. Developmental Cognitive Neuroscience, Special Section: The Developmental Neuroscience of Adolescence: Revisiting, Refining, and Extending Seminal Models, 17, 57–67.doi:10.1016/j.dcn.2015.10.010
Jones, A. W. (2019). Alcohol, its absorption, distribution, metabolism, and excretion in the body and pharmacokinetic calculations. WIREs Forensic Science, 1(5).doi:10.1002/wfs2.1340
Sarala, M., Miettunen, J., Koskela, J., Mustonen, A., Rose, R. J., Hurtig, T., Veijola, J., & Niemelä, S. (2020). Frequent intoxication and alcohol tolerance in adolescence: Associations with psychiatric disorders in young adulthood. Addiction, 115(5), 888–900.doi:10.1111/add.14889
Devi, A., Levin, M., & Waterhouse, A. L. (2023). Inhibition of ALDH2 by quercetin glucuronide suggests a new hypothesis to explain red wine headaches. Scientific Reports, 13(1).doi:10.1038/s41598-023-46203-y
Zhou, H., & Gelernter, J. (2024). Human genetics and epigenetics of alcohol use disorder. The Journal of Clinical Investigation, 134(16).doi:10.1172/JCI172885
Palmer, E., Tyacke, R., Sastre, M., Lingford-Hughes, A., Nutt, D., & Ward, R. J. (2019). Alcohol Hangover: Underlying Biochemical, Inflammatory and Neurochemical Mechanisms. Alcohol and Alcoholism, 54(3), 196–203.doi:10.1093/alcalc/agz016
Bereda, G. (2022). Mental and Physical Symptoms of Alcohol Hangover. Neurons and Neurological Disorders, 1(1), 1–4.doi:10.58489/2836-8851/004
Haass-Koffler, C. L., & Perciballi, R. (2020). Alcohol Tolerance in Human Laboratory Studies for Development of Medications to treat Alcohol Use Disorder. Alcohol and Alcoholism, 55(2), 129–135.doi:10.1093/alcalc/agz103
Obad, A., Peeran, A., Little, J. I., Haddad, G. E., & Tarzami, S. T. (2018). Alcohol-Mediated Organ Damages: Heart and Brain. Frontiers in Pharmacology, 9.doi:10.3389/fphar.2018.00081
Carvalho, A. F., Heilig, M., Perez, A., Probst, C., & Rehm, J. (2019). Alcohol use disorders. The Lancet, 394(10200), 781–792.doi:10.1016/S0140-6736(19)31775-1
Witkiewitz, K., Litten, R. Z., & Leggio, L. (2019). Advances in the science and treatment of alcohol use disorder. Science Advances, 5(9).doi:10.1126/sciadv.aax4043
Balloux, F., & van Dorp, L. (2017). Q&A: What are pathogens, and what have they done to and for us? BMC Biology, 15(1), 91.doi:10.1186/s12915-017-0433-z
Poon, M. M. L., & Farber, D. L. (2020). The Whole Body as the System in Systems Immunology. iScience, 23(9).doi:10.1016/j.isci.2020.101509
Sun, L., Su, Y., Jiao, A., Wang, X., & Zhang, B. (2023). T cells in health and disease. Signal Transduction and Targeted Therapy, 8(1), 235.doi:10.1038/s41392-023-01471-y
Zuluaga, P., Sanvisens, A., Teniente-Serra, A., El Ars, O., Fuster, D., Quirant-Sánchez, B., Martínez-Cáceres, E., & Muga, R. (2020). Loss of naive T lymphocytes is associated with advanced liver fibrosis in alcohol use disorder. Drug and Alcohol Dependence, 213.doi:10.1016/j.drugalcdep.2020.108046
Mace, E. M. (2023). Human natural killer cells: Form, function, and development. Journal of Allergy and Clinical Immunology, 151(2), 371–385.doi:10.1016/j.jaci.2022.09.022
Bottos, A., Gotthardt, D., Gill, J. W., Gattelli, A., Frei, A., Tzankov, A., Sexl, V., Wodnar-Filipowicz, A., & Hynes, N. E. (2016). Decreased NK-cell tumour immunosurveillance consequent to JAK inhibition enhances metastasis in breast cancer models. Nature Communications, 7(1).doi:10.1038/ncomms12258
Pascual, M., Calvo-Rodriguez, M., Núñez, L., Villalobos, C., Ureña, J., & Guerri, C. (2021). Toll-like receptors in neuroinflammation, neurodegeneration, and alcohol-induced brain damage. IUBMB Life, 73(7), 900–915.doi:10.1002/iub.2510
Caslin, B., Mohler, K., Thiagarajan, S., & Melamed, E. (2020). Alcohol as friend or foe in autoimmune diseases: A role for gut microbiome? Gut Microbes, 13(1).doi:10.1080/19490976.2021.1916278
Liu, M., Saredy, J., Zhang, R., Shao, Y., Sun, Y., Yang, W. Y., Wang, J., Liu, L., Drummer, C., Johnson, C., Saaoud, F., Lu, Y., Xu, K., Li, L., Wang, X., Jiang, X., Wang, H., & Yang, X. (2020). Approaching Inflammation Paradoxes—Proinflammatory Cytokine Blockages Induce Inflammatory Regulators. Frontiers in Immunology, 11.doi:10.3389/fimmu.2020.554301
El-Zayat, S. R., Sibaii, H., & Mannaa, F. A. (2019). Toll-like receptors activation, signaling, and targeting: An overview. Bulletin of the National Research Centre, 43(1), 187.doi:10.1186/s42269-019-0227-2
Gupta, S. C., Kunnumakkara, A. B., Aggarwal, S., & Aggarwal, B. B. (2018). Inflammation, a Double-Edge Sword for Cancer and Other Age-Related Diseases. Frontiers in Immunology, 9, 2160.doi:10.3389/fimmu.2018.02160
Chopade, P., Chopade, N., Zhao, Z., Mitragotri, S., Liao, R., & Chandran Suja, V. (2023). Alzheimer’s and Parkinson’s disease therapies in the clinic. Bioengineering & Translational Medicine, 8(1).doi:10.1002/btm2.10367
Çınaroğlu, M., Yılmazer, E., Ülker, S. V., Taçyıldız, K., & Tarlacı, S. (2025). Volumetric and cortical thickness alterations in alcohol dependence: Evidence of accelerated brain aging and clinical correlations. Frontiers in Psychiatry, 16.doi:10.3389/fpsyt.2025.1662842
Henmar, S., Simonsen, E. B., & Berg, R. W. (2020). What are the gray and white matter volumes of the human spinal cord? Journal of Neurophysiology, 124(6), 1792–1797.doi:10.1152/jn.00413.2020
Pereira-Rufino, L. da S., Gobbo, D. R., Conte, R., Sino, R. M. de, Oliveira, N. N. de, Fidalgo, T. M., Sato, J. R., Carrete Junior, H., Souza-Formigoni, M. L. O., Shi, Z., Vissoci, J. R. N., Wiers, C. E., & Céspedes, I. C. (2025). Reduced gray matter volume in limbic and cortical areas is associated with anxiety and depression in alcohol use disorder patients. Psychiatry Research: Neuroimaging, 347.doi:10.1016/j.pscychresns.2025.111946
Guggenmos, M., Schmack, K., Sekutowicz, M., Garbusow, M., Sebold, M., Sommer, C., Smolka, M. N., Wittchen, H.-U., Zimmermann, U. S., Heinz, A., & Sterzer, P. (2017). Quantitative neurobiological evidence for accelerated brain aging in alcohol dependence. Translational Psychiatry, 7(12), 1279.doi:10.1038/s41398-017-0037-y
Cole, J. H., & Franke, K. (2017). Predicting Age Using Neuroimaging: Innovative Brain Ageing Biomarkers. Trends in Neurosciences, 40(12), 681–690.doi:10.1016/j.tins.2017.10.001
Nutt, D., Hayes, A., Fonville, L., Zafar, R., Palmer, E. O. C., Paterson, L., & Lingford-Hughes, A. (2021). Alcohol and the Brain. Nutrients, 13(11), 3938.doi:10.3390/nu13113938
Herman, A. M., & Duka, T. (2019). Facets of impulsivity and alcohol use: What role do emotions play? Neuroscience & Biobehavioral Reviews, Addiction: A Neurobiological and Cognitive Brain Disorder, 106, 202–216.doi:10.1016/j.neubiorev.2018.08.011
Denier, N., Zahnd, K., Stein, M., Moggi, F., Kupper, Z., Federspiel, A., Wiest, R., Grieder, M., Soravia, L. M., & Bracht, T. (2025). Brain Connectivity of the Cingulate Cortex in Alcohol Use Disorder: Exploring Its Association With Mindfulness. Addiction Biology, 30(10).doi:10.1111/adb.70036
Stevens, B. W., Morris, J. K., Diazgranados, N., & Ramchandani, V. A. (2021). Common Gray Matter Reductions in Alcohol Use and Obsessive-Compulsive Disorders: A Meta-analysis. Biological Psychiatry Global Open Science, 2(4), 421–431.doi:10.1016/j.bpsgos.2021.11.010
Gröpper, S., Spengler, S., Stuke, H., Gawron, C. K., Parnack, J., Gutwinski, S., Wiers, C. E., & Bermpohl, F. (2016). Behavioral impulsivity mediates the relationship between decreased frontal gray matter volume and harmful alcohol drinking: A voxel-based morphometry study. Journal of Psychiatric Research, 83, 16–23.doi:10.1016/j.jpsychires.2016.08.006
Le Berre, A.-P., Fama, R., & Sullivan, E. V. (2017). Executive Functions, Memory, and Social Cognitive Deficits and Recovery in Chronic Alcoholism: A Critical Review to Inform Future Research. Alcoholism: Clinical and Experimental Research, 41(8), 1432–1443. doi:10.1111/acer.13431
Groh, J., & Simons, M. (2025). White matter aging and its impact on brain function. Neuron, 113(1), 127–139.doi:10.1016/j.neuron.2024.10.019
Daviet, R., Aydogan, G., Jagannathan, K., Spilka, N., Koellinger, P. D., Kranzler, H. R., Nave, G., & Wetherill, R. R. (2022). Associations between alcohol consumption and gray and white matter volumes in the UK Biobank. Nature Communications, 13(1), 1175. doi:10.1038/s41467-022-28735-5
Luo, L. (2021). Architectures of neuronal circuits. Science, 373(6559).doi:10.1126/science.abg7285
Pihlaja, R., Öhman, S., Ollila, H., Tuulio-Henriksson, A., Koskinen, S., Salmela, V., Tiainen, M., Hästbacka, J., & Hokkanen, L. (2026). Executive functioning after COVID-19: Associations between self-report, informant-report, objective cognition, and psychological factors. A prospective cohort study. Journal of Psychosomatic Research, 205.doi:10.1016/j.jpsychores.2026.112616
Peer, M., Nitzan, M., Bick, A. S., Levin, N., & Arzy, S. (2017). Evidence for Functional Networks within the Human Brain’s White Matter. Journal of Neuroscience, 37(27), 6394–6407.doi:10.1523/JNEUROSCI.3872-16.2017
Zahr, N. M. (2024). Alcohol Use Disorder and Dementia: A Review. Alcohol Research : Current Reviews, 44(1), 03. doi:10.35946/arcr.v44.1.03
Singer, E., Bhatt, K., Prashad, A., Rudman, L., Gadelmoula, I., & Michel, G. (2023). Diagnosis and Management of Marchiafava-Bignami Disease, a Rare Neurological Complication of Long-term Alcohol Abuse. Discoveries, 11(2).doi:10.15190/d.2023.7
De León Reyes, N. S., Bragg-Gonzalo, L., & Nieto, M. (2020). Development and plasticity of the corpus callosum. Development, 147(18).doi:10.1242/dev.189738
Teixeira, J., Pereira, I., Castanho, M., & Simões do Couto, F. (2025). What is the impact of thiamine deficiency on cognitive function in patients with alcohol use disorder? – A systematic review. European Journal of Internal Medicine, 134, 59–65.doi:10.1016/j.ejim.2025.01.008
Smith, T. J., Johnson, C. R., Koshy, R., Hess, S. Y., Qureshi, U. A., Mynak, M. L., & Fischer, P. R. (2021). Thiamine deficiency disorders: A clinical perspective. Annals of the New York Academy of Sciences, 1498(1), 9–28.doi:10.1111/nyas.14536
Li, P., Zhu, Z., Wang, Y., Zhang, X., Yang, C., Zhu, Y., Zhou, Z., Chao, Y., Long, Y., Gao, Y., Liu, S., Zhang, L., Gao, P., & Qu, Q. (2024). Substrate transport and drug interaction of human thiamine transporters SLC19A2/A3. Nature Communications, 15(1).doi:10.1038/s41467-024-55359-8
Bagash, H., Marwat, A., Marwat, A., Kraus, B., Bagash, H., Marwat, A., Marwat, A., & Kraus, B. (2021). A Case of Chronic Wernicke Encephalopathy (WE): An Underdiagnosed Phenomena. Cureus, 13.doi:10.7759/cureus.19100
Aloi, J., Crum, K. I., Blair, K. S., Zhang, R., Bashford-Largo, J., Bajaj, S., Schwartz, A., Carollo, E., Hwang, S., Leiker, E., Filbey, F. M., Averbeck, B. B., Dobbertin, M., & Blair, R. J. R. (2021). Individual associations of adolescent alcohol use disorder versus cannabis use disorder symptoms in neural prediction error signaling and the response to novelty. Developmental Cognitive Neuroscience, 48.doi:10.1016/j.dcn.2021.100944
Blandino, K., DiLeo, A., Antonoudiou, P., Weiss, G. L., & Maguire, J. (2025). Stress and alcohol impact network states involved in emotional processing: Relevance of comorbid AUD and psychiatric illnesses. Psychopharmacology.doi:10.1007/s00213-025-06928-2