SSRIs and Storks: The Complicated Relationship Between SSRIs and Pregnancy
Talia Mohideen
Illustrations by Maisy Richardson
Note: This article uses female-gendered language to refer to pregnant and postpartum people due to the vast majority of cited literature being focused on female-identifying subjects. The editors wish to acknowledge that pregnancy is independent of gender identity.
For many expecting mothers, pregnancy is filled with excitement about this new stage in life — starting a family and raising a child. However, pregnancy also brings new challenges, including new responsibilities, hormonal changes, and risks of various complications [1, 2, 3]. Women who take medication to manage depression and anxiety have the additional challenge of deciding whether to continue during pregnancy. Depression is a mood disorder characterized by persistent sadness, fatigue, changes in sleep or appetite, and a loss of interest or pleasure in hobbies; anxiety is a disorder characterized by fear, nervousness, and worry, as well as physical symptoms such as heart palpitations, shortness of breath, dizziness, and muscle tension [4, 5]. To cope with these conditions, more than thirty million Americans use antidepressants, with selective serotonin reuptake inhibitors (SSRIs) being the most common [6]. Initially introduced in the early 1980s, SSRIs such as Lexapro, Prozac, and Zoloft have become the standard pharmaceuticals used to treat depression over the past few decades [7]. This is largely due to their shorter list of associated side effects when compared with medications used earlier in psychiatric history, which carried a higher risk for cardiovascular issues, among other complications [8, 9]. However, as is common with most medications, SSRIs have their downsides. Common side effects of SSRIs include emotional blunting and sexual dysfunction [10, 11]. When women who take SSRIs become pregnant, they may have reason to be concerned about how this medication will impact their fetus — for example, potentially increasing the risk of miscarriage, preterm birth, and cardiac defects [12]. However, for women with depression, anxiety, or both, there are also risks of not using SSRIs during pregnancy [13, 14]. Women can become particularly vulnerable to depression during pregnancy if they are also coping with life stress and a lack of support [15]. Left untreated, depression jeopardizes maternal well-being by increasing the risk of suicidal ideation and postpartum depression (PPD) — conditions that pose a serious threat to both mother and fetus [12]. As a result, women are often faced with the difficult decision of whether to continue SSRI use during pregnancy.
Care Packages: How SSRIs Mitigate Depression and Anxiety Symptoms
An expectant mother experiencing depression may feel as though she is not living, but simply surviving [16]. When she gets out of bed in the morning, she may find it difficult to take her pregnancy vitamins or feel as though she has no purpose [16]. Watching her comfort TV show or chatting with friends, which used to bring her joy, makes her feel as though she is going through the motions without any emotion [17, 18]. Anxiety disorders are similarly incapacitating: for a mother with anxiety, her thoughts can be consuming [19]. She might worry about everything — things she did or said, the health of her fetus, and her family’s future. It feels impossible to stop these thoughts from occupying her mind, making it nearly impossible to focus [19]. With millions enduring the reality of depression and anxiety every day, it begs the question of how and why the disorders develop [20, 21]. What are the neurological mechanisms behind these conditions? And why do SSRIs provide a way to mitigate such difficult thoughts and feelings?
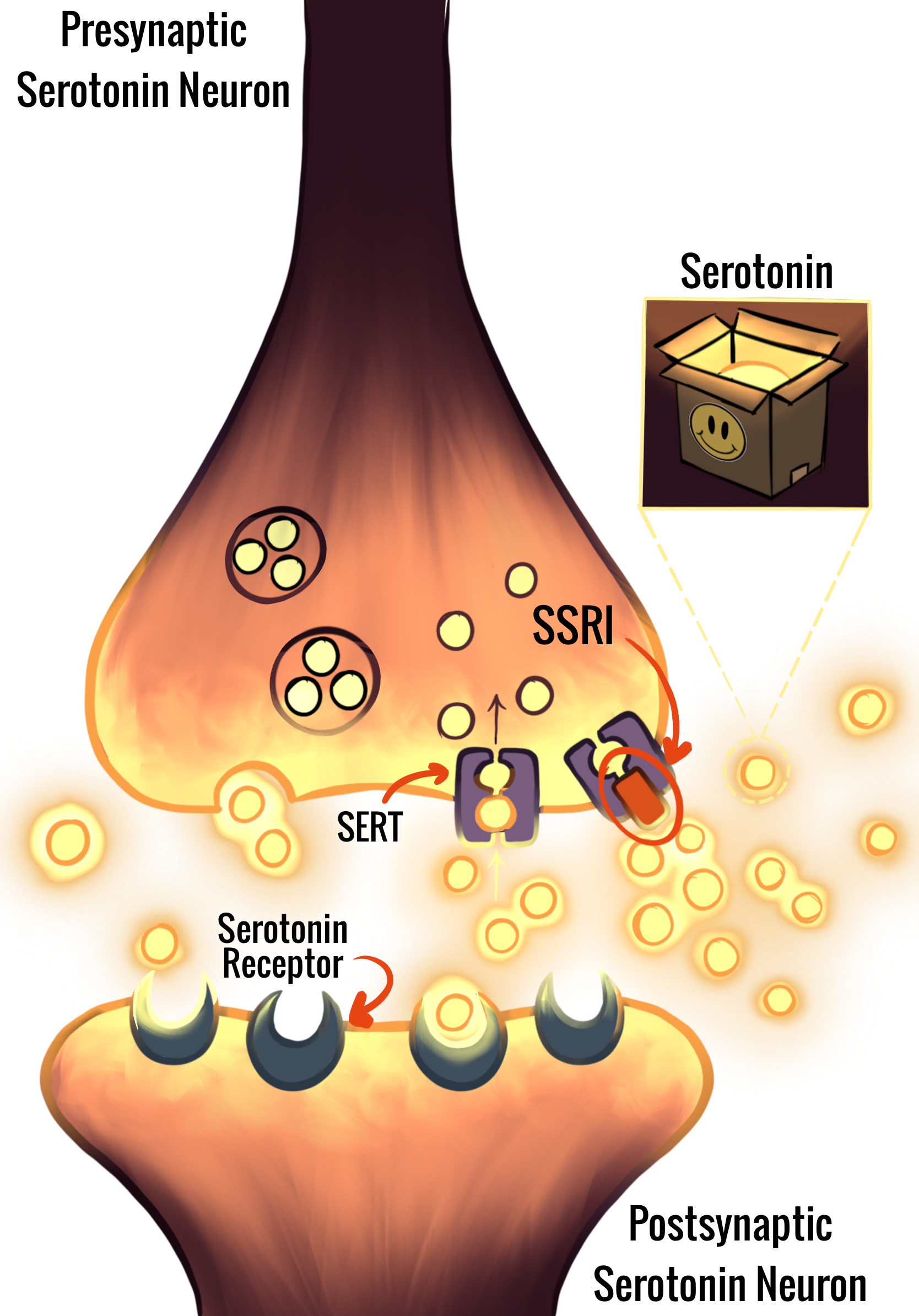
Imagine that the main communication cells of your brain, called neurons, are houses in a neighborhood expecting a package delivery. These packages, or neurotransmitters, are molecules that carry messages from one neuron to the next [22, 23]. In order to do this, neurotransmitters must cross the synapse, the gap between two neurons, and bind to receptors that specifically recognize that neurotransmitter [22, 23]. Afterwards, the process of reuptake allows neurotransmitters to be reabsorbed into the neuron that released them, just as when mail goes back to the sender [24, 25]. Serotonin is a neurotransmitter involved in regulating mood, sleep, and appetite [26, 27]. Increased serotonin in the synapse, which makes receptor binding more likely, is linked to improved emotional states — when serotonin ‘letters’ enter someone’s synaptic ‘mailbox’ regularly, they may experience more positive emotions [26]. Sometimes, however, the mailbox is completely empty or overflowing with packages, both of which can be harmful, just as both deficient and excessive synaptic serotonin can be problematic [28, 29]. An empty mailbox may be the result of excessive reuptake. The flooding of serotonin into the synapse and its subsequent reuptake must therefore be balanced [24, 25].
Serotonin deficiency in the synapse, which is associated with depression and anxiety, may be due to dysregulation of the serotonin transporter (SERT), which results in excessive reuptake [30, 31]. In this case, dysregulated SERT is analogous to a mailman forgetting to deliver any packages to the houses and simply bringing them all back to the post office. SSRIs bind to and inhibit SERT, reducing serotonin reuptake, so the packages are delivered and stay in the mailbox for longer until they can be received or taken back to the post office [32, 33, 34]. The serotonin molecules stay in the synapse longer, resulting in increased and prolonged signaling [32, 33, 34]. In other words, SSRIs allow the ‘positive packages’ to continue to their destination rather than being taken up by SERT.
A Bun in the Oven: Serotonin and SSRIs in Embryonic Development
Serotonin and SSRIs may cross the placenta — an organ that provides the fetus with water and nutrients crucial to its survival and development [35, 36, 37]. Serotonin, which can act as both a neurotransmitter and a hormone, is crucial for supporting embryonic development [38]. Hormones are molecules that carry chemical signals through the blood, similar to how neurotransmitters carry messages between neurons [39]. During pregnancy, serotonin is involved in the positioning of organs, eye development, and the production of neurons in the fetus. SSRIs may block serotonin absorption in the placenta, thereby affecting aspects of fetal development that are dependent on the concentration of serotonin. As a result, taking SSRIs during pregnancy increases the risk of birth defects such as heart, brain, and skull malformations. In summary, SSRI use by the mother can have a myriad of effects on the child, both during development and after birth, contributing to controversy about whether women should continue their regular SSRI treatment while pregnant [14, 36].
Lost in Transit: SSRI Withdrawal and Postpartum Depression
While SSRIs can be helpful to people with depression and anxiety, there is also cause for an individual deciding to discontinue medication [40, 41]. Emotional blunting, a decrease in emotional responsiveness that is often described as feeling ‘numb,’ is a common side effect of SSRIs that causes many people to consider stopping or changing their medication regimen [40]. When SSRIs increase the amount of serotonin in the synapse, the brain physically adapts. Specifically, it is believed that the SSRIs cause serotonin receptors to become less sensitive and decrease in number, leading to less serotonin binding overall [42]. When people discontinue SSRIs, reuptake is no longer inhibited, and the concentration of serotonin at the synapse decreases. As a result, emotional blunting ceases, and the reversion to stronger emotions can be difficult to manage [43]. This decrease in serotonin and in receptor sensitivity is what underlies SSRI withdrawal, a phenomenon characterized by sleep disruption, irritability, dizziness, nausea, anxiety, and depression [42]. Other withdrawal effects include struggles with concentration and maintaining an active social life [43]. Withdrawal symptoms will continue until the brain returns to its original level of serotonin sensitivity, which can take as short as two weeks or as long as several months after stopping SSRI usage [41, 44].
SSRI withdrawal can be even more challenging as a result of the significant changes to expectant mothers’ bodies and brains as hormones fluctuate [12, 45, 46]. The increased levels of the hormones oxytocin and estrogen during pregnancy regulate the release of other hormones and neurotransmitters that are involved in motherhood [47, 48]. For example, oxytocin is involved in breastfeeding, which is important in facilitating attachment between mother and child [49]. Additionally, the hormonal fluctuations that occur during pregnancy can have a major impact on a pregnant person’s mental state — for example, increased estrogen is associated with increased vulnerability to depression [50, 51]. The decision to discontinue SSRI treatment alone can have a major impact on mood, and discontinuation during pregnancy can have even more profound effects [12, 45, 46]. Depression symptoms, such as poor sleep and nutrition, which may re-emerge during SSRI withdrawal, can increase the risk of other health issues for both the mother and fetus [52, 53]. Thus, pregnant individuals who decide to discontinue SSRIs may benefit from other forms of treatment for depression or anxiety, such as cognitive or psychotherapy, or tapering off these drugs [12].
Mothers in the postpartum period are at risk for developing depression due to major hormonal changes [54]. Thus, not taking SSRIs can also be dangerous for mothers who previously used them, as it could lead to postpartum depression (PPD), a specific type of depression affecting mothers after giving birth [12]. Nearly 13-19% of new mothers experience PPD, which is characterized by depressed mood, sleep and appetite disturbance, fatigue, and feeling guilty, worthless, and overwhelmed [55, 56, 57]. Individuals struggling with this condition may be overly worried or preoccupied about their infant’s health, feeding, and bathing safety, to the point of dysfunction. In extreme cases, they may even experience suicidal thoughts and thoughts of harming their child. PPD is a very complex disorder, hence the seemingly contradictory symptoms [57]. Children of mothers suffering from PPD can also be affected, as they may experience impairments in cognitive development, as well as emotional and behavioral issues [58]. SSRI treatment for mothers with PPD can mitigate some of their symptoms [58]. Coupled with unclear information on depression and SSRI treatment options, further research is needed to address the mental health concerns affecting the pregnant and postpartum population.
A Complicated Relationship: Pre-eclampsia and SSRIs
There is a lack of consensus regarding whether SSRIs increase or decrease the risk of certain health conditions, prompting discussions regarding whether it is best to continue or discontinue SSRIs [6]. For example, pre-eclampsia, a condition characterized by high blood pressure in expectant mothers, is a leading cause of maternal morbidity and mortality [2, 59, 60]. Mothers diagnosed with this condition may experience headaches, visual disturbances, chest pain, or confusion [61]. Pre-eclampsia increases the risk of fetal death and preterm birth [62]. Additionally, children born to mothers with pre-eclampsia tend to be underweight due to preterm birth, and thus may have a higher risk of cardiovascular and metabolic complications later in life [62, 63]. If left untreated, pre-eclampsia can progress into eclampsia, a life-threatening condition [64]. A mother with eclampsia will experience one or more seizures, which involve strong and sustained convulsions of several muscles, inability to breathe, and increased heart rate [65, 66].
The connection between SSRIs and pre-eclampsia is complicated. There is a potential bidirectional relationship between depression and pre-eclampsia — pregnant women diagnosed with pre-eclampsia are more than twice as likely to develop mood and anxiety disorders [67]. Depression symptoms themselves may contribute to a lack of self-care, which can then increase the risk of pre-eclampsia development due to poor maternal health overall. There may be a greater risk of pre-eclampsia associated with SSRI use; however, the validity of this relationship is contested [6]. Since there are shared determinants between depression and pre-eclampsia, individuals taking SSRIs may be at higher risk [6]. It is also plausible that SSRIs prevent pre-eclampsia [68]. Increased serotonin is associated with decreased arginine vasopressin (AVP), a hormone associated with pre-eclampsia. By decreasing AVP, SSRIs may decrease the risk of pre-eclampsia [68]. The case of pre-eclampsia is not an isolated example, and it illustrates the myriad challenges associated with choosing to continue SSRIs during pregnancy [67].
Troubles Upon Arrival: Infant Withdrawal
Approximately one third of infants exposed to SSRIs during the third trimester show signs of Poor Neonatal Adaptation Syndrome (PNAS) — a condition that, like SSRI withdrawal, is characterized by irritability, muscle tone irregularities, jitteriness, and feeding difficulties [69, 70]. These symptoms have been observed in infants several hours after birth; however, these are typically mild and often resolve naturally over several days or weeks [71]. In acute cases, PNAS may involve more severe symptoms such as respiratory issues and seizures [72]. The effects of PNAS can be reduced through breastfeeding, skin-to-skin contact, and pharmacotherapy. Since SSRIs can be passed into breastmilk, breastfeeding can also help the infant gradually taper off the medication and reduce withdrawal symptoms [14]. Some mothers may choose to discontinue SSRI treatment during pregnancy due to fear of their child experiencing PNAS; however, in most cases, the risks associated with this condition seem to be minor [71].
Priority (Fe)mail
In recent years, there has been an increase in the popularity of political movements against psychiatric medications, as well as greater misinformation and weaponization of maternal and women’s health [73, 74]. While skepticism is important in science, we as a society must also recognize the benefits modern medicine provides. Millions of people benefit from SSRIs, and for most, the small risks associated with continuing them during pregnancy are minute compared to those of discontinuation [6, 12, 52]. Of course, each individual is different and must make their own decision in consultation with professionals [12]. Future research will likely give us a better understanding of how continued and discontinued SSRI use during pregnancy affects maternal and fetal well-being. The mother’s and fetus’s health are deeply intertwined — a healthy mother ensures a healthy environment for the fetus to develop. But, above all, the mother’s own health must be prioritized, as an improved quality of life allows for a more fulfilling experience both as a parent and as an individual.
Reference
1. Jee, S. B., & Sawal, A. (2024). Physiological changes in pregnant women due to hormonal changes. Cureus,16(3), e55544. doi:10.7759/cureus.55544
2. Álvarez-Fernández, I., Prieto, B., & Álvarez, F. V. (2016). Preeclampsia. Revista del Laboratorio Clínico, 9(2), 81-89. doi:10.1016/j.labcli.2016.04.002
3. Stewart, D. E., & Vigod, S. N. (2019). Postpartum depression: pathophysiology, treatment, and emerging Therapeutics. Annual Review of Medicine, 70, 183-196. doi:10.1146/annurev-med-041217-011106
4. Sarno, E., Moeser, A. J., & Robison, A. J. (2021). Neuroimmunology of depression. Advances in Pharmacology 91, 259–292. doi:10.1016/bs.apha.2021.03.004
5. Szuhany, K.L., & Simon, N. M. (2022). Anxiety disorders: A review. JAMA328(24), 2431–2445. doi:10.1001/jama.2022.22744
6. Gumusoglu, S. B., Schickling, B. M., Vignato, J. A., Santillan, D. A., & Santillan, M. K. (2022). Selective serotonin reuptake inhibitors and preeclampsia: A quality assessment and meta-analysis. Pregnancy Hypertension, 30, 36-43. doi:10.1016/j.preghy.2022.08.001
7. Fagiolini, A., Cuomo, A., & McIntyre, R. S. (2024). SSRIs. In Pocket guide to practical psychopharmacology: SSRIs and SNRIs in clinical practice 7–75. Springer Nature. doi:10.1007/978-3-031-80490-8_2
8. Cowen, P. J. (2023). SSRIs in the treatment of depression: A pharmacological cul-de-sac? In M. Browning, P. J. Cowen, & T. Sharp (Eds.), Emerging neurobiology of antidepressant treatments, 66, 1–19. Springer Nature.doi:10.1007/7854_2023_447
9. Wang, S.-M., Han, C., Bahk, W.-M., Lee, S.-J., Patkar, A. A., Masand, P. S., & Pae, C.-U. (2018). Addressing the side effects of contemporary antidepressant drugs: A comprehensive review. Chonnam Medical Journal, 54(2), 101–112.doi:10.4068/cmj.2018.54.2.101
10. Camino, S., Strejilevich, S. A., Godoy, A., Smith, J., & Szmulewicz, A. (2023). Are all antidepressants the same? The consumer has a point. Psychological Medicine, 53(9), 4004–4011. doi:10.1017/S0033291722000678
11. Jing, E. & Straw-Wilson, K. (2016). Sexual dysfunction in selective serotonin reuptake inhibitors (SSRIs) and potential solutions: A narrative literature review. Mental Health Clinician, 6(4), 191–196.doi:0.9740/mhc.2016.07.191
12. Alwan, S., Friedman, J.M. & Chambers, C. (2016). Safety of selective serotonin reuptake inhibitors in pregnancy: A review of current evidence. CNS Drugs 30, 499–515 https://doi.org/10.1007/s40263-016-0338-3
13. Bérard, A., Sheehy, O., Zhao, J. P., Vinet, É., Bernatsky, S., & Abrahamowicz, M. (2017). SSRI and SNRI use during pregnancy and the risk of persistent pulmonary hypertension of the newborn. British Journal of Clinical Pharmacology, 83(5), 1126-1133. doi:10.1111/bcp.13194
14. Dubovicky, M., Belovicova, K., Csatlosova, K. & Bogi, E. (2017). Risks of using SSRI / SNRI antidepressants during pregnancy and lactation. Interdisciplinary Toxicology, 10(1), 30-34. doi:10.1515/intox-2017-0004
15. Jahan, N., Went, T. R., Sultan, W., Sapkota, A., Khurshid, H., Qureshi, I. A., & Alfonso, M. (2021). Untreated depression during pregnancy and its effect on pregnancy outcomes: A systematic review. Cureus, 13(8), e17251. doi:10.7759/cureus.17251
16. Watson, R., Harvey, K., McCabe, C., & Reynolds, S. (2019). Understanding anhedonia: A qualitative study exploring loss of interest and pleasure in adolescent depression. European Child & Adolescent Psychiatry, 29(4), 489–499. doi:10.1007/s00787-019-01364-y
17. Serretti, A. (2023). Anhedonia and depressive disorders. Clinical Psychopharmacology and Neuroscience, 21(3), 401. doi:10.9758/cpn.23.1086
18. Tolentino, J. C., & Schmidt, S. L. (2018). DSM-5 criteria and depression severity: Implications for clinical practice. Frontiers in Psychiatry, 9, 450. doi:10.3389/fpsyt.2018.00450
19. Adwas, A. A., Jbireal, J. M., & Azab, A. E. (2019). Anxiety: Insights into signs, symptoms, etiology, pathophysiology, and treatment. East African Scholars Journal of Medical Sciences, 2(10), 580–591. doi:10.36349/EASMS.2019.v02i10.006
20. Remes, O., Brayne, C., & Lafortune, L. (2016). A systematic review of reviews on the prevalence of anxiety disorders in adult populations. Brain and Behavior, 6(7), e00497. doi:10.1002/brb3.497
21. Stringaris, A. (2017). Editorial: What is depression? Journal of Child Psychology and Psychiatry, 58(12), 1287-1289. doi:10.1111/jcpp.12844
22. Südhof, T. C. (2018). Towards an understanding of synapse formation. Neuron, 100(6), 276–293. doi:10.1016/j.neuron.2018.08.042
23. Hampel, L., Lau, T. (2022). Neurobiological principles: Neurotransmitters. In: Riederer, P., Laux, G., Nagatsu, T., Le, W., Riederer, C. (Eds.), NeuroPsychopharmacotherapy. Springer Nature. doi:10.1007/978-3-030-62059-2_365
24. Özçete, Ö. D., Banerjee, A., & Kaeser, P. S. (2024). Mechanisms of neuromodulatory volume transmission. Molecular Psychiatry, 29(11), 3680-3693. doi:10.1038/s41380-024-02608-3
25. Zeppelin, T., Ladefoged, L. K., Sinning, S., Periole, X., & Schiøtt, B. (2018). A direct interaction of cholesterol with the dopamine transporter prevents its out-to-inward transition. PLOS Computational Biology, 14(1), e1005907. doi:10.1371/journal.pcbi.1005907
26. Jones, L. A., Sun, E. W., Martin, A. M., & Keating, D. J. (2020). The ever-changing roles of serotonin. The International Journal of Biochemistry & Cell Biology, 125, 105776. doi:10.1016/j.biocel.2020.105776
27. Pourhamzeh, M., Moravej, F. G., Arabi, M., Shahriari, E., Mehrabi, S., Ward, R., Ahadi, R., & Joghataei, M. T. (2022). The roles of serotonin in neuropsychiatric disorders. Cellular and Molecular Neurobiology, 42(6), 1671–1692. doi:10.1007/s10571-021-01064-9
28. Kayabaşı, Y., Güneş, B., Erbaş, O. (2021). Serotonin Receptors and Depression. Journal of Experimental and Basic Medical Sciences, 2(2), 240-246. doi:10.5606/jebms.2021.75662
29. Mikkelsen, N., Damkier, P., & Pedersen, S. A. (2023). Serotonin syndrome—A focused review. Basic & Clinical Pharmacology & Toxicology, 133(2), 124-129. doi:10.1111/bcpt.13912
30. Carhart-Harris, R., & Nutt, D. (2017). Serotonin and brain function: A tale of two receptors. Journal of Psychopharmacology 31(9), 1091. doi:10.1177/0269881117725915
31. Ye, X., Ghosh, S., Shin, B. C., Ganguly, A., Maggiotto, L., Jacobs, J. P., & Devaskar, S. U. (2024). Brain serotonin and serotonin transporter expression in male and female postnatal rat offspring in response to perturbed early life dietary exposures. Frontiers in Neuroscience, 18, 1363094. doi:10.3389/fnins.2024.1363094
32. Murthy, M. K. (2025). Molecular pathways linking the serotonin transporters (SERT) to depressive disorder: From mechanisms to treatments. Neuroscience, 584, 2-31. doi:10.1016/j.neuroscience.2025.08.009
33. Sharp, T., Collins, H. (2023). Mechanisms of SSRI therapy and discontinuation. In: Browning, M., Cowen, P.J., Sharp, T. (Eds.) Emerging Neurobiology of Antidepressant Treatments. Current Topics in Behavioral Neurosciences, 66. Springer Nature. doi:10.1007/7854_2023_452
34. Xue, W., Wang, P., Li, B., Li, Y., Xu, X., Yang, F., Yao, X., Chen, Y. Z., Xu, F., & Zhu, F. (2016). Identification of the inhibitory mechanism of FDA approved selective serotonin reuptake inhibitors: An insight from molecular dynamics simulation study. Physical Chemistry Chemical Physics, 18(5), 3260–3271. doi:10.1039/C5CP05771J
35. Aengenheister, L., Keevend, K., Muoth, C., Schönenberger, R., Diener, L., Wick, P., & Buerki-Thurnherr, T. (2018). An advanced human in vitro co-culture model for translocation studies across the placental barrier. Scientific Reports, 8(1), 5388. doi:10.1038/s41598-018-23410-6
36. Bérard, A., Levin, M., Sadler, T., & Healy, D. (2019). Selective Serotonin Reuptake Inhibitor Use During Pregnancy and Major Malformations: The Importance of Serotonin for Embryonic Development and the Effect of Serotonin Inhibition on the Occurrence of Malformations. Bioelectricity, 1(1), 18-29. doi:10.1089_bioe.2018.0003
37. Roberts, R. M., Green, J. A., & Schulz, L. C. (2016). The evolution of the placenta. Reproduction, 152(5), R179-R189. doi:10.1530/REP-16-0325
38. Lv, J., & Liu, F. (2017). The role of serotonin beyond the central nervous system during embryogenesis. Frontiers in Cellular Neuroscience, 11, 259363. doi:10.3389/fncel.2017.00074
39. Stárka, L., & Dušková, M. (2020). What Is a Hormone? Physiological Research, 69(Suppl 2), S183. doi:10.33549/physiolres.934509
40. Masdrakis, V. G., Markianos, M., & Baldwin, D. S. (2023). Apathy associated with antidepressant drugs: a systematic review. Acta Neuropsychiatrica, 35(4), 189–204. doi:10.1017/neu.2023.6
41. Palmer, E. G., Sornalingam, S., Page, L., & Cooper, M. (2023). Withdrawing from SSRI antidepressants: Advice for primary care. The British Journal of General Practice, 73(728), 138. doi:10.3399/bjgp23X732273
42. Horowitz, M. A., & Taylor, D. (2019). Tapering of SSRI treatment to mitigate withdrawal symptoms. The Lancet Psychiatry, 6(6), 538–546. doi:10.1016/S2215-0366(19)30032-X
43. Mahmood, R., Wallace, V., Wiles, N., Kessler, D., Button, K. S., & Fairchild, G. (2024). The lived experience of withdrawal from selective serotonin reuptake inhibitor (SSRI) antidepressants: A qualitative interview study. Health Expectations, 27(1), e13966. doi:10.1111/hex.13966
44. Davies, J., & Read, J. (2019). A systematic review into the incidence, severity and duration of antidepressant withdrawal effects: Are guidelines evidence-based? Addictive Behaviors, 97, 111-121. doi:10.1016/j.addbeh.2018.08.027
45. Servin-Barthet, C., Martínez-García, M., Pretus, C., Paternina-Die, M., Soler, A., Khymenets, O., Pozo, Ó. J., Leuner, B., Vilarroya, O., & Carmona, S. (2023). The transition to motherhood: Linking hormones, brain and behaviour. Nature Reviews Neuroscience, 24(10), 605-619. doi:10.1038/s41583-023-00733-6
46. Jee, S. B., & Sawal, A. (2024). Physiological changes in pregnant women due to hormonal changes. Cureus, 16(3), e55544. doi:10.7759/cureus.55544
47. Liu, N., Yang, H., Han, L., & Ma, M. (2022). Oxytocin in women’s health and disease. Frontiers in Endocrinology, 13, 786271. doi:10.3389/fendo.2022.786271
48. Berkane, N., Liere, P., Oudinet, J. P., Hertig, A., Lefèvre, G., Pluchino, N., Schumacher, M., & Chabbert-Buffet, N. (2017). From pregnancy to preeclampsia: A key role for estrogens. Endocrine Reviews, 38(2), 123-144. doi:10.1210/er.2016-1065
49. Kim, S., & Strathearn, L. (2016). Oxytocin and maternal brain plasticity. New Directions for Child and Adolescent Development, 2016(153), 59-72. doi:10.1002/cad.20170
50. Trifu, S., Vladuți, A., & Popescu, A. (2019). Neuroendocrine aspects of pregnancy and postpartum depression. Acta Endocrinologica (Bucharest), 15(3), 410–415. doi:10.4183/aeb.2019.410
51. Albert, K. M., & Newhouse, P. A. (2019). Estrogen, stress, and depression: Cognitive and biological interactions. Annual Review of Clinical Psychology, 15, 399-423. doi:10.1146/annurev-clinpsy-050718-095557
52. Liu, X., Molenaar, N., Agerbo, E., Momen, N. C., Rommel, A.-S., Lupattelli, A., Bergink, V., & Munk-Olsen, T. (2022). Antidepressant discontinuation before or during pregnancy and risk of psychiatric emergency in Denmark: A population-based propensity score–matched cohort study. PLOS Medicine, 19(1), e1003895. doi:10.1371/journal.pmed.1003895
53. Henssler, J., Schmidt, Y., Schmidt, U., Schwarzer, G., Bschor, T., & Baethge, C. (2024). Incidence of antidepressant discontinuation symptoms: A systematic review and meta-analysis. The Lancet Psychiatry, 11(7), 526-535. doi:10.1016/S2215-0366(24)00133-0
54. Wu, X., & Jin, R. (2025). Effects of postpartum hormonal changes on the immune system and their role in recovery. Acta Biochimica Polonica, 72, 14241. doi:10.3389/abp.2025.14241
55. Suryawanshi, O. I. V., & Pajai, S. (2022). A comprehensive review on postpartum depression. Cureus, 14(12), e32745. doi:10.7759/cureus.32745
56. Emerson, M. R., Mathews, T. L., Struwe, L. (2018). Postpartum Depression Screening for New Mothers at Well Child Visits. The American Journal of Maternal/Child Nursing, 43(3), 139-145, doi:10.1097/NMC.0000000000000426
57. Stewart, D. E., & Vigod, S. N. (2019). Postpartum depression: Pathophysiology, treatment, and emerging therapeutics. Annual Review of Medicine, 70, 183-196. doi:10.1146/annurev-med-041217-011106
58. Liu C, Ystrom E, McAdams TA. (2023). Long-term maternal and child outcomes following postnatal SSRI treatment. JAMA Network Open, 6(8), e2331270. doi:10.1001/jamanetworkopen.2023.31270
59. Kattah, A. (2020). Preeclampsia and kidney disease: Deciphering cause and effect. Current Hypertension Reports, 22(11), 91. doi:10.1007/s11906-020-01099-1
60. Dines, V., Šuvakov, S., Kattah, A., Vermunt, J., Narang, K., Jayachandran, M., Garovic, V. D., & others. (2023). Preeclampsia and the kidney: Pathophysiology and clinical implications. Comprehensive Physiology, 13(1), 4231–4267.doi:10.1002/j.2040-4603.2023.tb00249.x
61. Vigil-De Gracia, P., Vargas, C., Sánchez, J., Collantes-Cubas, J. (2023). Preeclampsia: Narrative review for clinical use. Heliyon, 9(3), e14187. doi:10.1016/j.heliyon.2023.e14187
62. Bokslag, A., Van Weissenbruch, M., Mol, B. W., & De Groot, C. J. (2016). Preeclampsia: Short and long-term consequences for mother and neonate. Early Human Development, 102, 47-50. doi:10.1016/j.earlhumdev.2016.09.007
63. Zhu, W., Wang, Q., & Chen, M. (2025). Identifying genes and traits associated with pre-eclampsia using summary statistics. PLOS One, 20(5), e0323683. doi:10.1371/journal.pone.0323683
64. Ahmed, A., Rezai, H., & Broadway-Stringer, S. (2017). Evidence-based revised view of the pathophysiology of preeclampsia. In M. S. Islam (Ed.), Hypertension: From basic research to clinical practice 956, 355–374. Springer International Publishing. doi:10.1007/5584_2016_168
65. Fishel Bartal, M., & Sibai, B. M. (2020). Eclampsia in the 21st century. American Journal of Obstetrics and Gynecology, 226(2 Supp.), S1239–S1247. doi:10.1016/j.ajog.2020.09.037
66. Nass, R. D., Zur, B., Elger, C. E., Holdenrieder, S., & Surges, R. (2019). Acute metabolic effects of tonic-clonic seizures. Epilepsia Open, 4(4), 599-608. doi:10.1002/epi4.12364
67. Zhao, C., Chen, F., Li, Q., Peng, L., & Yue, C. (2025). Bidirectional Mendelian randomization analysis of mood disorders and risk of preeclampsia-eclampsia. International Journal of Women’s Health, 17, 3557–3565. doi:10.2147/IJWH.S539218
68. Vignato, J. A., Gumusoglu, S. B., Davis, H. A., Scroggins, S. M., Hamilton, W. S., Brandt, D. S., Pierce, G. L., Knosp, B. A., Santillan, D. A., & Santillan, M. K. (2023). Selective serotonin reuptake inhibitor use in pregnancy and protective mechanisms in preeclampsia. Reproductive Sciences, 30(2), 701–712. doi:10.1007/s43032-022-01065-z
69. Evans, S. H., Cameron, M. W., & Burton, J. . (2017). Hypertonia. Current Problems in Pediatric and Adolescent Health Care, 47(7), 161-166. doi:10.1016/j.cppeds.2017.06.005
70. Viguera, A. C., McElheny, S. A., Caplin, P. S., Kobylski, L. A., Rossa, E. T., Young, A. V., Gaccione, P., Góez-Mogollón, L., Freeman, M. P., & Cohen, L. S. (2023). Risk of poor neonatal adaptation syndrome among infants exposed to second-generation atypical antipsychotics compared to antidepressants: Results from the National Pregnancy Registry for Psychiatric Medications. Journal of Clinical Psychiatry, 84(1), 22m14492. doi:10.4088/JCP.22m14492
71. Cantin, C., Snelgrove-Clarke, E., Gaudet, L., & Ross-White, A. (2024). Characteristics of breastfeeding newborns in the first month of life with in utero selective serotonin reuptake inhibitor medication exposure: A scoping review protocol. JBI Evidence Synthesis, 22(1), 144–152. doi:10.11124/JBIES-23-00048
72. Convertino, I., Sansone, A. C., Marino, A., Galiulo, M. T., Mantarro, S., Antonioli, L., Fornai, M., Blandizzi, C., & Tuccori, M. (2016). Neonatal adaptation issues after maternal exposure to prescription drugs: Withdrawal syndromes and residual pharmacological effects. Drug Safety, 39(10), 903–924. doi:10.1007/s40264-016-0435-8
73. Öngür D. (2025). Psychiatry and the Make America Healthy Again Commission. JAMA Network, 333(24), 2145–2146. doi:10.1001/jama.2025.6255
74. Gostin, L. O., Finch, A., & Lurie, P. (2025). Making America healthy again: Remedies for Robert F. Kennedy Jr.'s campaign against chronic disease. Hastings Center Report, 55(4), 2-8. doi:10.1002/hast.5020