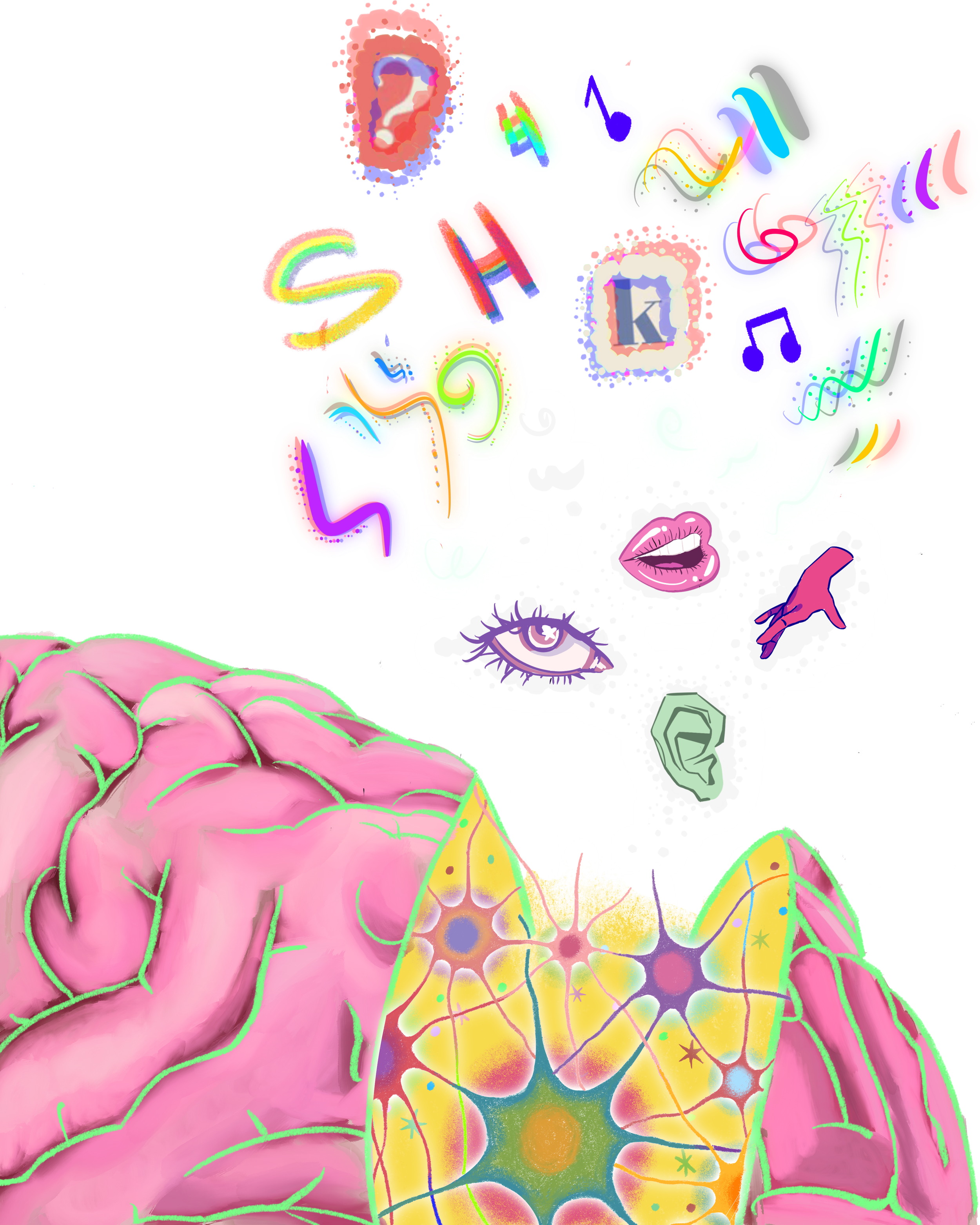
All Senses to Center Stage: Synesthesia in Action
Tara Dacey
Illustrations by Alexandra Tapia
Most people have a favorite song and a favorite color, but for a person with synesthesia, these could be one and the same. Synesthesia is the involuntary perception of additional sensations that occurs when a stimulus, such as an image or sound, simultaneously activates two or more typically unrelated senses [1, 2]. Synesthetic experiences manifest as combinations of sight, hearing, touch, taste, or smell [3, 4]. For a sensory experience to be considered synesthesia, it must clearly be the result of a stimulus, like automatically perceiving the color pink at the sound of a flute [1, 5]. Such instances are distinct from hallucinations, which are not triggered by external stimuli [6]. While synesthesia may seem unusual or almost psychedelic, it should not be mistaken for an indication of other mental conditions or a cause of distress. Rather, synesthesia is a benign and unique variation of sensory perception [6].
About 4% of the global population experiences synesthesia, meaning that at a concert with 10,000 people in attendance, roughly 400 people might see colors or visualize words in response to the music, lighting, or other stimuli present [4]. Manifestations vary widely among individuals with synesthesia; different people experience unique sensations in response to identical stimuli [2, 7]. At the strum of a guitar, one person with synesthesia might taste a mango, whereas another might physically feel the smooth texture of the music gliding across their hands. However, synesthetic experiences remain relatively stable for each person: the same stimulus consistently elicits the same response throughout their life [4, 8]. For example, if a person with synesthesia hears the music note ‘A’ at age seven and perceives the color magenta, they will still visualize magenta upon hearing the same note at age seventy-five. The direction of synesthetic perception also tends to remain stable [4]. In most cases, perception is unidirectional: the inducer, the triggering stimulus, leads to the concurrent, the additional perceptual event, and not the other way around.. For the person who visualizes magenta alongside the music note ‘A,’ sound induces the experience of color, but a magenta-colored stimulus would be unlikely to induce the sound of the music note ‘A’. The unidirectionality and lifetime consistency of synesthesia make it easier to categorize and understand [4].
Cast List: Causes and Clusters
While synesthesia is considered a single condition, each specific combination of inducer and concurrent is its own subtype, typically named based on the stimulus and the additional sensory experience elicited [1,2,4]. Although each subtype of synesthesia has unique characteristics and manifests differently in every person, subtypes can be grouped into clusters based on a shared concurrent experience [2,9]. For example, the cluster created for colored sequences includes the number-color and weekday-color synesthesia subtypes [2]. Clusters are helpful tools for understanding differences in synesthetic experiences, but there is currently no overarching theory explaining how the condition develops [2]. However, the existence of shared clusters among people supports the theory that some subtypes may stem from a common cause [1, 2]. Multiple subtypes of synesthesia are associated with global hyperconnectivity, which refers to abnormally heightened communication between brain regions [10, 11]. Networks between regions are formed through neuroplasticity, the ability of brain cells called neurons to create or strengthen connections with other neurons [4]. Brain-derived neurotrophic factor (BDNF), a protein involved in the development of neuron structure and function, plays a key role in facilitating neuroplasticity [4]. Concentrations of the protein are generally higher in people with synesthesia than in those without, which could support the theory that global hyperconnectivity and increased neuroplasticity are associated with the development of synesthesia [1, 4, 12].
People with synesthesia display heightened connectivity between regions of the brain responsible for sensory processing, including parietal, visual, and auditory regions [11, 13]. The parietal cortex is crucial for integrating information from multiple senses to create a cohesive perception of a stimulus [14]. The disinhibited feedback model provides one theory of how and why synesthesia manifests, explaining it through the involvement of the parietal cortex [13, 15]. In most people, information is sent from brain areas involved in initial sensory processing to the parietal cortex, not the other way around. By contrast, in people with synesthesia, the parietal cortex may transfer sensory information back to the areas involved in initial sensory processing, causing concurrent experiences [13, 15]. It is hypothesized that the feedback connections from the parietal cortex are disinhibited, which is similar to releasing the brakes of a car [13]. Pressing the brakes inhibits the car’s movement, causing it to come to a stop. Likewise, by inhibiting the parietal cortex, feedback connections are seemingly blocked, and synesthetic experiences may lessen as a result [13]. Disinhibition occurs when you lift your foot off the brake, taking the car out of an inhibited state and allowing it to move forward. If the parietal cortex is disinhibited in synesthesia, the feedback connections might be abnormally powerful and thereby activate additional sensory modalities: visual, auditory, tactile, gustatory (taste), and olfactory (smell) [13, 16]. The activation of sensory areas consequently elicits a concurrent [13, 16].
Behind the Scenes: Brain Structure and Genes
In addition to the possible structural and functional mechanisms underlying synesthetic experiences, genetics may also play a role in the development of the condition [9, 17]. Close relatives of people with synesthesia are more likely to also have the condition [9, 17]. Members of the same family may even develop different subtypes of synesthesia, further indicating that the condition has a common origin regardless of which synesthetic experiences manifest [9]. However, no single gene is responsible for the occurrence of synesthesia; instead, multiple genes may act as predisposing factors [9,17]. Even if someone does not have synesthesia but has relatives with the condition, they may have greater connectivity and BDNF concentrations than a person who does not have relatives with synesthesia [18]. While BDNF levels differ between people with and without synesthesia, differences in concentration alone cannot explain why people with synesthesia have such vastly distinct experiences [1]. Relatives of people with synesthesia may also display increased creativity and improved episodic memory, traits frequently observed in people with synesthesia, without necessarily experiencing synesthesia themselves [18]. The presence of shared cognitive traits between those with synesthesia and their relatives without synesthesia supports the possibility of a heritable factor of synesthesia [2]. If a specific genetic component of synesthesia is identified, it could explain why certain people are prone to developing synesthesia [19].
In combination with genetics, atypical brain structures seen in early childhood development may lead to the emergence of synesthesia [1, 20]. Environmental influences could be crucial in determining whether someone who is predisposed to synesthesia through brain structure develops the condition, as well as how it manifests [2, 18]. People who develop synesthesia might observe an object that involves multiple senses, such as a blue toy that plays music, and then internalize and enhance the association [2]. The mechanism behind this internalization is based on the adaptability of infants’ brains, which contain over 100 billion neurons. As learning occurs, the pathways the brain uses most often grow stronger, while the less useful ones fade away through a process known as ‘pruning’ [21]. In people with developmental synesthesia, a lack of pruning leads to excessive connections after early phase development, causing hyperconnectivity [1]. As a result, developmental synesthesia emerges in early childhood and continues through adulthood [1]. In contrast, post-accidental synesthesia is a subtype that typically arises due to permanent nerve damage caused by brain injury [20, 22]. For example, in people who have experienced damage to vision-processing regions, a form of visual synesthesia might occur as an attempt by the brain to overcompensate for the loss of sight and better interpret sensory stimuli [20]. Regardless of whether changes occur during development or after injury, structural abnormalities are implicated in the onset of synesthesia [1, 20].
Roll Call: Subtypes of Synesthesia
There are seemingly endless ways for the brain to alter its processing of stimuli; more than 80 subtypes of synesthesia have been discovered thus far [4]. One of synesthesia’s most common subtypes is grapheme-color (GC) synesthesia, named after graphemes — letters or numbers — that trigger the perception of color [8, 23]. For the 1–2% of the population living with GC synesthesia, atypically strong associations between graphemes and colors may form during childhood [4, 23]. People with GC synesthesia exhibit increased brain activity in the color-processing region in response to grapheme stimuli, possibly leading them to perceive certain characters as colors [4]. The brain region that responds to graphemes, known as the visual word form area (VWFA), and the brain region responsible for color processing are adjacent [1, 11]. When a grapheme is processed in the VWFA of someone with synesthesia, the color-processing region may also be activated: this is an example of the cross-activation model [11]. Similarly, it has been hypothesized that enhanced multisensory integration — the brain’s process of unifying sensory information from multiple modalities to create a single perception — may also be rooted in the hyperconnectivity of the VWFA and color-processing area [11, 24, 25]. Those with GC synesthesia sometimes display enhanced recall of facts through long-term memory and a more vivid recall of personal details, known as episodic memory [2, 23]. The basis of this enhanced memory may lie in the tendency to think using mental images, as well as in the hyperconnectivity seen in those with synesthesia [2].
Another subtype involving the VWFA is ticker-tape synesthesia (TTS), in which hearing speech results in seeing the words on a banner in the mind’s eye, separate from the external world [13, 26]. A person with TTS could hear someone say, ‘Let’s get brunch on Saturday!’ and automatically visualize the sentence as if it were torn from a magazine. TTS is the only known subtype in which the inducer and concurrent are both language-based; language-processing centers in the brain overactivate the VWFA, which transforms heard speech into internally visualized letters [13, 26, 27]. As with other synesthesia subtypes, individuals with TTS may have an enhanced working memory, a form of short-term memory that allows information to be stored and manipulated, similar to a mental workspace [27]. An enhanced working memory could mean a person with synesthesia exhibits advanced strategic thinking while planning moves during a game of chess or possesses an above-average ability to remember names [27].
While some subtypes of synesthesia, like TTS, have a single predominant theory, auditory-visual (AV) synesthesia has many proposed mechanisms [11]. AV synesthesia is a subtype characterized by an auditory stimulus that causes a visual, typically colorful, experience. The neurological processes that lead a person with AV synesthesia to, say, hear a cat’s meow and perceive the color turquoise may be similar to those underlying GC synesthesia. Specifically, cross-activation between parietal, auditory, and color-processing areas may play a role in AV synesthesia. However, the directionality of signal transmission between the regions activated by the inducer, concurrent areas, and the parietal cortex remains undetermined. Two models currently predominate: the two-stage model and the disinhibited feedback model. The two-stage model suggests that signal transmission travels from the inducer areas to concurrent areas and finally to the parietal cortex. Alternatively, the disinhibited feedback model posits that transmission stems from the inducer areas to the parietal cortex and then to concurrent areas. Recall how the rate of communication from the parietal cortex can be like the movement of a car when you lift your foot off the brakes. Understanding the abnormal pathway taken by stimuli to higher processing areas in AV synesthesia is vital in determining whether the two-step or disinhibited feedback model is correct, though both theories suggest that this subtype results from abnormal parietal cortex involvement during sensory transmission [11].
Set Design: Synesthesia and Creativity
Many explanations exist for how synesthesia develops in individuals, but some theories additionally suggest that the condition may be evolutionarily advantageous for the human species in general [18, 29]. Indeed, synesthesia may enhance individuals’ abilities to understand stimuli and unconsciously seek additional clues for identification through other senses [18, 29]. Individuals with synesthesia can exhibit cognitive abnormalities, including heightened memory, intellectual curiosity, and creativity [1, 2]. The ability to interpret and create requires cross-sensory mapping, a process closely related to synesthesia [30]. Cross-sensory mapping is the correspondence between sensory modalities — like matching higher pitches to increased brightness — which is distinct from combining information from multiple senses through multisensory integration [14, 31]. Making associations between senses requires abstract thinking, a process that also supports the understanding of figurative language, such as metaphors [30]. Metaphors begin with sensory perception, often combining multiple sensory modalities. For example, the meaning of the term ‘sour face’ is understood through the connection of the gustatory and visual modalities. Individuals with heightened creativity have a greater ability to establish metaphorical connections and to link seemingly unrelated concepts [30]. During creative processes, correspondence between the senses is increased for all people, but in people with synesthesia, hyperconnectivity and cross-activation between modalities connect the senses even further [11, 30, 32]. People with synesthesia may also have more vivid mental imagery than those without, further aiding creativity and allowing them to derive more enjoyment from imaginative activities [30]. Picture a potter with synesthesia wishing to craft a tea set, with a tea cup resembling a pink lotus in full bloom and a saucer like a lily pad. The potter may be able to visualize the lotus cup and lily pad saucer in such vivid detail that the mental image serves as the perfect reference for crafting the set. While synesthesia does not guarantee greater creativity, it prompts unique sensory experiences that can fuel artistic and creative endeavors.
Double Feature: Synesthesia and Other Conditions
Just as no two pieces of original art are identical, no two individuals have the same brain. Unique individual patterns of brain development may be responsible for certain subtypes of synesthesia, similar to other neurological conditions [4]. People with autism spectrum disorder (ASD) display a higher prevalence of synesthesia than the general population, and people with synesthesia tend to exhibit more common characteristics of autism than the general population, supporting a potential link between the conditions [33]. ASD and synesthesia share many cognitive attributes, including hypersensitivity to stimuli, enhanced perceptual functioning, increased attention to detail, and superior memory [17, 34]. Evidence for the connection includes global hyperconnectivity, which is often present in both people with ASD and people with synesthesia [34, 35]. Additionally, for people with ASD, synesthesia has been suggested to contribute to the development of savant abilities, such as perfect pitch or extraordinary mathematical capacity, which are present in about 10% of people with ASD [36, 37]. There is a positive correlation between genes associated with ASD and those associated with synesthesia, but because each condition is influenced by many genes, it is difficult to pinpoint which ones may influence either condition [17].
Synesthesia may also be related to autonomous sensory meridian responses (ASMR), pleasant tingling sensations that initiate at the scalp and are typically triggered by whispering and crisp sounds [32]. Inducers for ASMR responders are also common inducers for people with synesthesia [32]. The crisp sound of a snare drum may cause an ASMR responder to feel tingling sensations and a person with sound-taste synesthesia to taste black licorice [34]. Synesthesia tends to be more prevalent in ASMR responders than in the general population, and people with synesthesia are more likely to be ASMR responders [32]. The high frequency of overlap between people who respond to ASMR and those with synesthesia may be explained by similar mechanisms. Both conditions involve highly individualized experiences regarding inducers and concurrents, as well as multisensory experiences. Additionally, ASMR responses might result from the cross-activation of the primary cortex and regions related to touch, which parallels the cross-activation model of synesthesia. Both people with synesthesia and ASMR responders exhibit abnormal patterns of functional connectivity, supporting the theory of shared developmental origins. Furthermore, the argument has been made that ASMR could be classified as its own subtype of synesthesia [32]!
Pulling Back the Curtain: Why continued research is important
As a form of sensory perception, synesthesia could provide valuable insight into how the brain works. Determining whether the structure of specific brain regions predisposes a person to synesthesia is not about identifying the ‘magic’ key that makes color out of music, but understanding how slightly different brains lead to incredibly different states of awareness [18]. Perhaps what is special about being human is that, though we generally share similar internal machinery, there are countless minute differences that define our individual selves and experiences. For those with the condition, synesthetic perception impacts not just baseline sensory information, but may also continuously influence how one understands and connects to the world. The highly individualized expressions of synesthesia make large-scale studies challenging, but further research could expand the existing understandings of the connections between sensory modalities and neuroplasticity, in both synesthesia itself and other conditions [4]. Our brains are inherently flexible, and synesthesia is just one of many intriguing manifestations.
References
Ward, J., Simner, J., Simpson, I., Rae, C., del Rio, M., Eccles, J. A., & Racey, C. (2024). Synesthesia is linked to large and extensive differences in brain structure and function as determined by whole-brain biomarkers derived from the HCP (human connectome project) cortical parcellation approach. Cerebral Cortex, 34(11). doi:10.1093/cercor/bhae446
Ward, J., & Simner, J. (2020). Chapter 13 - Synesthesia: the current state of the field. Multisensory Perception, 283–300. doi:10.1016/b978-0-12-812492-5.00013-9
Bianchi, I., Paradis, C., & van de Weijer, J. (2025). Perceptual structure of opposites across sensory modalities. Language and Cognition, 17. doi:10.1017/langcog.2025.10016
Eckardt, N., Sinke, C., Bleich, S., Lichtinghagen, R., & Zedler, M. (2024). Investigation of the relationship between neuroplasticity and grapheme-color synesthesia. Frontiers in Neuroscience, 18. doi:10.3389/fnins.2024.1434309
Sun, Y., Yang, H., Zhu, D., Wang, Y., & Suen, R. (2026). Harnessing synesthesia to unlock new pathways in learning for students with disabilities. Learning and Instruction, 102, 102292. doi:10.1016/j.learninstruc.2025.102292
Collerton, D., Barnes, J., Diederich, N. J., Dudley, R., ffytche, D., Friston, K., Goetz, C. G., Goldman, J. G., Jardri, R., Kulisevsky, J., Lewis, S. J. G., Nara, S., O’Callaghan, C., Onofrj, M., Pagonabarraga, J., Parr, T., Shine, J. M., Stebbins, G., Taylor, J.-P., … Weil, R. S. (2023). Understanding visual hallucinations: A new synthesis. Neuroscience & Biobehavioral Reviews, 150, 105208. doi:10.1016/j.neubiorev.2023.105208
Berger, J. J., Harris, I. M., Whittingham, K. M., Terpening, Z., & Watson, J. D. (2019). Substantiating synesthesia: A novel aid in a case of grapheme-colour synesthesia and concomitant dyscalculia. Neurocase, 26(1), 29–35. doi:10.1080/13554794.2019.1695846
Chu, I., Kokash, A., Li, S. P., & Vendrame, M. (2025). Grapheme-color synesthesia in patients with epilepsy: A pilot study. Epilepsy & Behavior, 166, 110378. doi:10.1016/j.yebeh.2025.110378
Tilot, A. K., Kucera, K. S., Vino, A., Asher, J. E., Baron-Cohen, S., & Fisher, S. E. (2018). Rare variants in axonogenesis genes connect three families with sound–color synesthesia. Proceedings of the National Academy of Sciences, 115(12), 3168–3173. doi:10.1073/pnas.1715492115
Hillary, F. G., & Grafman, J. H. (2017). Injured brains and adaptive networks: The benefits and costs of hyperconnectivity. Trends in Cognitive Sciences, 21(5), 385–401. doi:10.1016/j.tics.2017.03.003
Brauchli, C., Elmer, S., Rogenmoser, L., Burkhard, A., & Jäncke, L. (2017). Top–down signal transmission and global hyperconnectivity in auditory‐visual synesthesia: Evidence from a functional EEG resting‐state study. Human Brain Mapping, 39(1), 522–531. doi:10.1002/hbm.23861
Mueller, K., Arelin, K., Möller, H. E., Sacher, J., Kratzsch, J., Luck, T., Riedel-Heller, S., Villringer, A., & Schroeter, M. L. (2016). Serum BDNF correlates with connectivity in the (pre)motor hub in the aging human brain—a resting-state fmri pilot study. Neurobiology of Aging, 38, 181–187. doi:10.1016/j.neurobiolaging.2015.11.003
Hauw, F., Béranger, B., & Cohen, L. (2024). Subtitled speech: The neural mechanisms of ticker-tape synaesthesia. Brain, 147(7), 2530–2541. doi:10.1093/brain/awae114
Coslett, B. H., & Schwartz, F. M. (2018). Chapter 18 - The parietal lobe and language. (2018). Handbook of Clinical Neurology, 365–375. doi:10.1016/b978-0-444-63622-5.00018-8
Lalwani, P., & Brang, D. (2019). Stochastic resonance model of synaesthesia. Philosophical Transactions of the Royal Society B: Biological Sciences, 374(1787), 20190029. doi:10.1098/rstb.2019.0029
Yeo, B. B., & Yu, J. (2025). Atypical cortical regional homogeneity observed in grapheme-colour synaesthesia and its subtypes. Cortex, 192, 135–151. doi:10.1016/j.cortex.2025.09.003
Bouvet, L., Amsellem, F., Maruani, A., Tonus-Vic Dupont, A., Mathieu, A., Bourgeron, T., Delorme, R., & Mottron, L. (2019). Synesthesia & autistic features in a large family: Evidence for spatial imagery as a common factor. Behavioural Brain Research, 362, 266–272. doi:10.1016/j.bbr.2019.01.014
Ward, J., & Filiz, G. (2020). Synaesthesia is linked to a distinctive and heritable cognitive profile. Cortex, 126, 134–140. doi:10.1016/j.cortex.2020.01.002
Speed, L. J., Croijmans, I., Dolscheid, S., & Majid, A. (2021). Crossmodal associations with olfactory, auditory, and tactile stimuli in children and adults. I-Perception, 12(6). doi:10.1177/20416695211048513
Yong, Z., Hsieh, PJ. & Milea, D. Seeing the sound after visual loss: functional MRI in acquired auditory-visual synesthesia. Exp Brain Res 235, 415–420 (2017). doi:10.1007/s00221-016-4802-6
Sakai, J. (2020). How synaptic pruning shapes neural wiring during development and, possibly, in disease. Proceedings of the National Academy of Sciences, 117(28), 16096–16099. doi:10.1073/pnas.2010281117
Schwartzman, D. J., Bor, D., Rothen, N., & Seth, A. K. (2019). Neurophenomenology of induced and natural synaesthesia. Philosophical Transactions of the Royal Society B: Biological Sciences, 374(1787), 20190030. doi:10.1098/rstb.2019.0030
Anash, S., & Boileau, A. (2024). Grapheme-color synesthesia and its connection to memory. Cureus. doi:10.7759/cureus.67524
Choi, I., & Lee, S.-H. (2025). Locomotion-dependent auditory gating to the parietal cortex guides multisensory decisions. Nature Communications, 16(1). doi:10.1038/s41467-025-57347-y
Newell, F. N., & Mitchell, K. J. (2016). Multisensory integration and cross-modal learning in synaesthesia: A unifying model. Neuropsychologia, 88, 140–150. doi:10.1016/j.neuropsychologia.2015.07.026
Delsanti, R., Hauw, F., Lahbari, R., Bouhali, F., & Cohen, L. (2025). Bridging speech and Sight: White matter anatomy in ticker-tape synaesthesia. Brain Communications, 7(5). doi:10.1093/braincomms/fcaf316
Hauw, F., El Soudany, M., Rosso, C., Daunizeau, J., & Cohen, L. (2023). A single case neuroimaging study of tickertape synesthesia. Scientific Reports, 13(1). doi:10.1038/s41598-023-39276-2
Hauw, F., El Soudany, M., & Cohen, L. (2023). The advantage of being a synesthete: The behavioral benefits of ticker-tape synesthesia. Cortex, 168, 226–234. doi:10.1016/j.cortex.2023.08.011
Chun, C. A., & Hupé, J. (2015). Are synesthetes exceptional beyond their synesthetic associations? A systematic comparison of creativity, personality, cognition, and mental imagery in synesthetes and controls. British Journal of Psychology, 107(3), 397–418. doi:10.1111/bjop.12146
Merter, S. (2017). Synesthetic approach in the design process for enhanced creativity and multisensory experiences. The Design Journal, 20(sup1), S4519-S4528. doi:10.1080/14606925.2017.1352948
Liu, Q., & Lupyan, G. (2026). The unity of sense and mind: A review of cross-domain mapping. Psychonomic Bulletin & Review, 33(1). doi:10.3758/s13423-025-02805-3
Poerio, G. L., Ueda, M., & Kondo, H. M. (2022). Similar but different: High prevalence of synesthesia in autonomous sensory meridian response (ASMR). Frontiers in Psychology, 13. doi:10.3389/fpsyg.2022.990565
Hughes, J. E. A., Simner, J., Baron-Cohen, S., Treffert, D. A., & Ward, J. (2017). Is synaesthesia more prevalent in autism spectrum conditions? Only where there is prodigious talent. Multisensory Research, 30(3–5), 391–408. doi:10.1163/22134808-00002558
van Leeuwen, T. M., Neufeld, J., Hughes, J., & Ward, J. (2020). Synaesthesia and autism: Different developmental outcomes from overlapping mechanisms? Cognitive Neuropsychology, 37(7–8), 433–449. doi:10.1080/02643294.2020.1808455
Limon, A., & Corona-Moreno, M. (2025). Hyperconnectivity and disrupted signal-to-noise processing in autism. The Palgrave Encyclopedia of Disability, 1–9. doi:10.1007/978-3-031-40858-8_179-1
van Leeuwen, T. M., Wilsson, L., Norrman, H. N., Dingemanse, M., Bölte, S., & Neufeld, J. (2021). Perceptual processing links autism and synesthesia: A co-twin control study. Cortex, 145, 236–249. doi:10.1016/j.cortex.2021.09.016
Rudziński, G., Pożarowska, K., Brzuszkiewicz, K., & Soroka, E. (2024). An outline of savant syndrome. Psychiatria Polska, 58(4), 681–691. doi:10.12740/pp/onlinefirst/157104